QUESTION IMAGE
Question
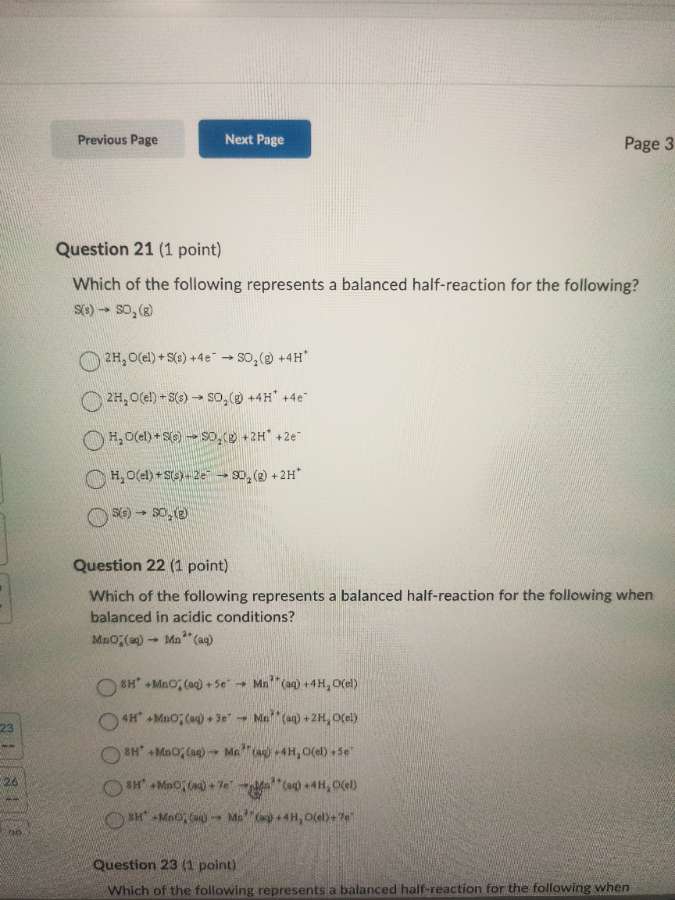
question 21 (1 point)
which of the following represents a balanced half - reaction for the following?
s(s) → so₂(g)
2h₂o(l) + s(s) + 4e⁻ → so₂(g) + 4h⁺
2h₂o(l) + s(s) → so₂(g) + 4h⁺ + 4e⁻
h₂o(l) + s(s) → so₂(g) + 2h⁺ + 2e⁻
h₂o(l) + s(s) + 2e⁻ → so₂(g) + 2h⁺
s(s) → so₂(g)
question 22 (1 point)
which of the following represents a balanced half - reaction for the following when balanced in acidic conditions?
mno₄⁻(aq) → mn²⁺(aq)
8h⁺ + mno₄⁻(aq) + 5e⁻ → mn²⁺(aq) + 4h₂o(l)
4h⁺ + mno₄⁻(aq) + 3e⁻ → mn²⁺(aq) + 2h₂o(l)
8h⁺ + mno₄⁻(aq) → mn²⁺(aq) + 4h₂o(l) + 5e⁻
8h⁺ + mno₄⁻(aq) + 7e⁻ → mn²⁺(aq) + 4h₂o(l)
8h⁺ + mno₄⁻(aq) → mn²⁺(aq) + 4h₂o(l) + 7e⁻
question 23 (1 point)
which of the following represents a balanced half - reaction for the following when
Question 21
Step1: Analyze oxidation state change
S in \( S(s) \) has oxidation state 0, in \( SO_2(g) \) it is +4. So S is oxidized, loses electrons.
Step2: Balance O with \( H_2O \)
To balance O in \( S \to SO_2 \), add 2 \( H_2O \) on left: \( 2H_2O + S \to SO_2 \)
Step3: Balance H with \( H^+ \)
Now balance H: 4 \( H^+ \) on right (since 2 \( H_2O \) has 4 H).
Step4: Balance charge
Left: charge from S (0) + \( H_2O \) (neutral) = 0. Right: \( SO_2 \) (neutral) + 4 \( H^+ \) (+4) = +4. So need to lose 4 \( e^- \) (oxidation, electrons on right). So reaction: \( 2H_2O(l) + S(s) \to SO_2(g) + 4H^+ + 4e^- \)
Step1: Analyze oxidation state change
Mn in \( MnO_4^- \) is +7, in \( Mn^{2+} \) is +2. So Mn is reduced, gains 5 \( e^- \).
Step2: Balance O with \( H_2O \)
\( MnO_4^- \to Mn^{2+} \): add 4 \( H_2O \) on right to balance O (4 O in \( MnO_4^- \)).
Step3: Balance H with \( H^+ \)
Add 8 \( H^+ \) on left (4 \( H_2O \) has 8 H).
Step4: Balance charge
Left: \( 8H^+ \) (+8) + \( MnO_4^- \) (-1) = +7. Right: \( Mn^{2+} \) (+2) + 4 \( H_2O \) (neutral) = +2. So need to gain 5 \( e^- \) (reduction, electrons on left). Reaction: \( 8H^+ + MnO_4^- + 5e^- \to Mn^{2+} + 4H_2O \)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\( 2H_2O(l) + S(s) \to SO_2(g) + 4H^+ + 4e^- \) (the second option)