QUESTION IMAGE
Question
question 33 of 49
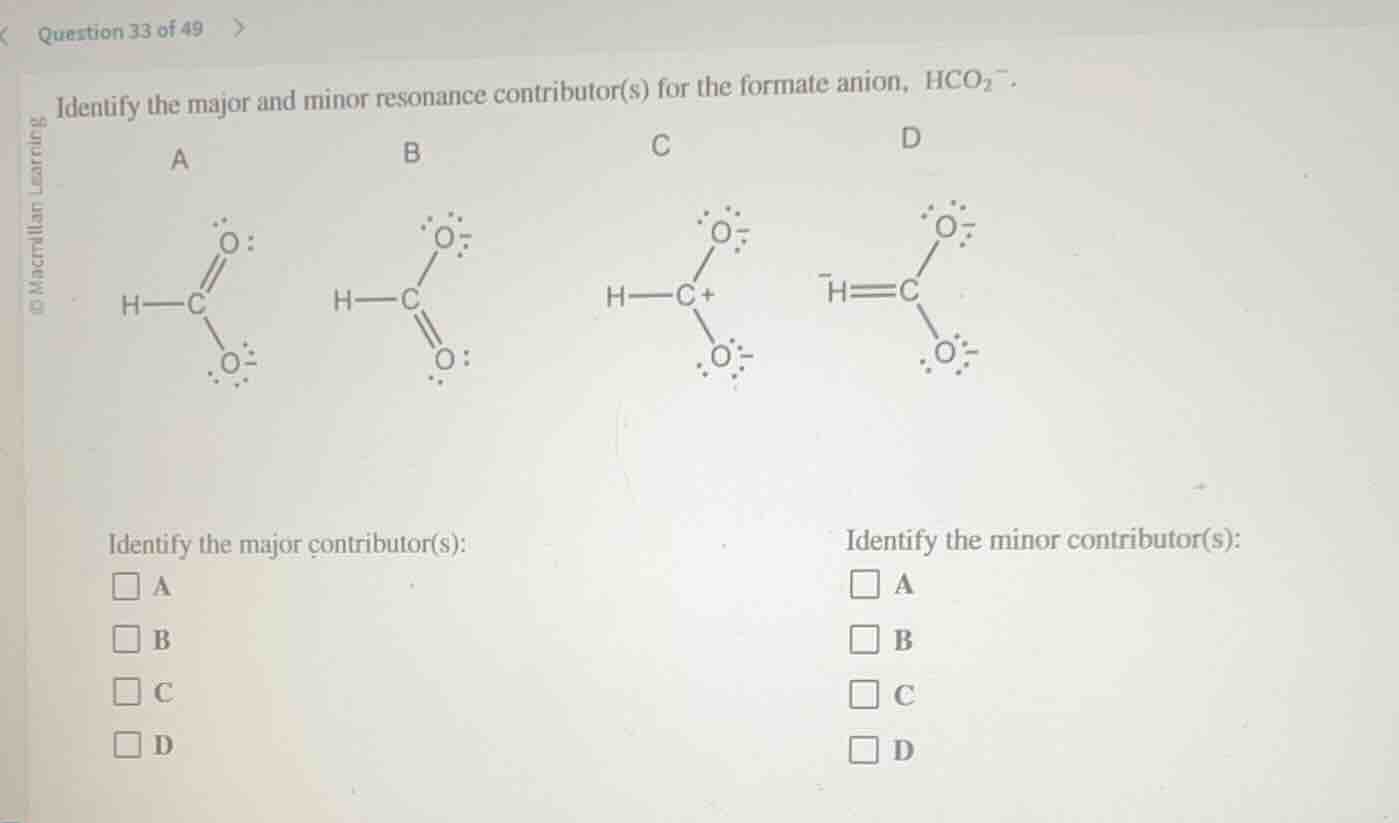
identify the major and minor resonance contributor(s) for the formate anion, hco₂⁻.
a
h—c
o:
o⁻
b
h—c
o⁻
o:
c
h—c⁺
o⁻
o⁻
d
⁻h═c
o⁻
o⁻
identify the major contributor(s):
☐ a
☐ b
☐ c
☐ d
identify the minor contributor(s):
☐ a
☐ b
☐ c
☐ d
Major resonance contributors are the most stable structures: they have full octets for all atoms, minimal formal charge separation, and negative charges on electronegative atoms. Structures A and B meet these criteria, with full octets, negative charges on oxygen (electronegative), and are equivalent in stability.
Minor contributors are less stable: Structure C has a positively charged carbon (electron-deficient, no full octet) and charge separation, making it less stable. Structure D has a negatively charged hydrogen, which is highly unstable as hydrogen cannot bear a negative charge, so it is a very minor contributor, but still a resonance form.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Identify the major contributor(s):
A. $\ce{H-C(=O)-O^{-}}$ (structure A), B. $\ce{H-C(=O^{-})-O}$ (structure B)
Identify the minor contributor(s):
C. $\ce{H-C^{+}(-O^{-})-O^{-}}$ (structure C), D. $\ce{^{-}H=C(-O^{-})-O^{-}}$ (structure D)