QUESTION IMAGE
Question
question 40 of 49
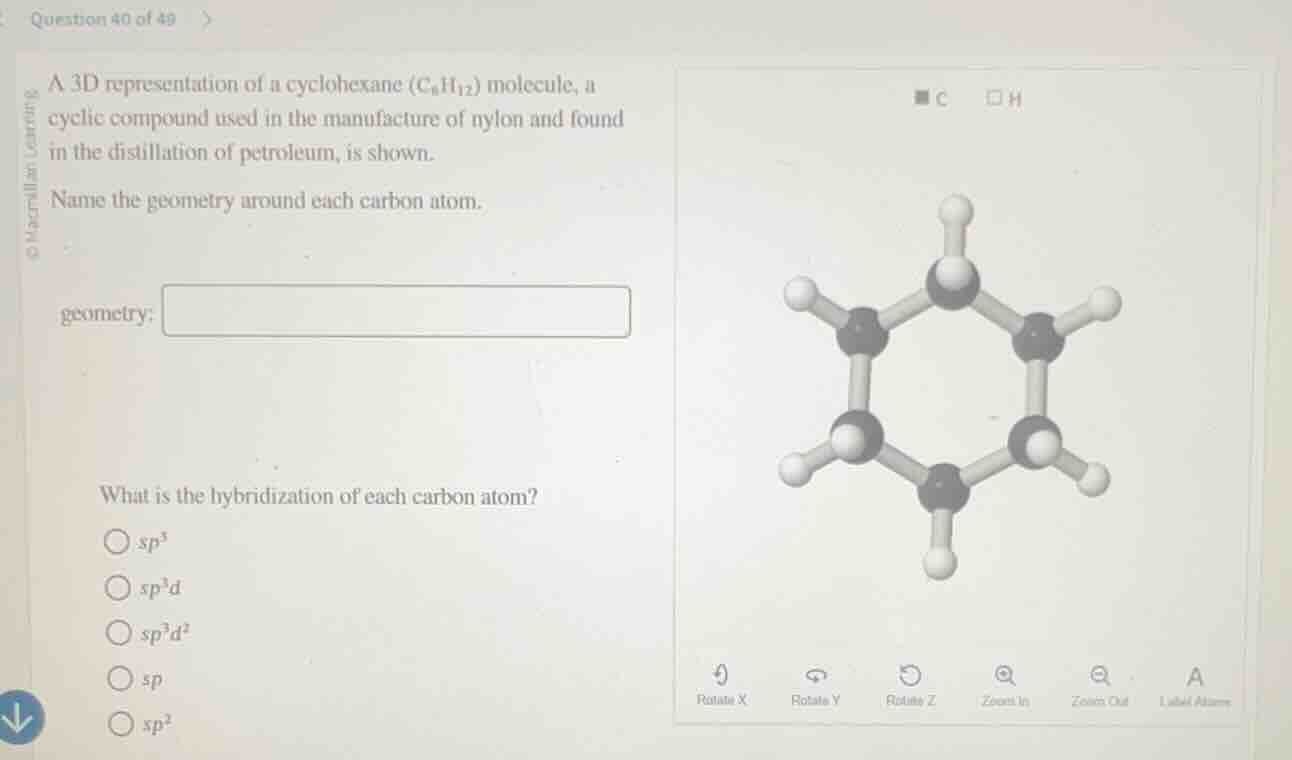
a 3d representation of a cyclohexane (c₆h₁₂) molecule, a cyclic compound used in the manufacture of nylon and found in the distillation of petroleum, is shown.
name the geometry around each carbon atom.
geometry:
what is the hybridization of each carbon atom?
○ sp³
○ sp³d
○ sp³d²
○ sp
○ sp²
(image of cyclohexane molecule with c and h labeled, and rotation/zoom options)
Step1: Analyze carbon bonding
Each carbon in cyclohexane ($\text{C}_6\text{H}_{12}$) forms 4 single bonds: 2 with adjacent carbon atoms and 2 with hydrogen atoms, with no lone electron pairs on the carbon atoms.
Step2: Relate bonding to geometry
A central atom with 4 bonding electron domains and 0 lone pairs has a tetrahedral electron domain and molecular geometry, as predicted by VSEPR theory.
Step3: Relate bonding to hybridization
An atom with 4 bonding electron domains uses $sp^3$ hybridization, where one $s$ orbital and three $p$ orbitals combine to form 4 equivalent hybrid orbitals for single bonding.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
geometry: tetrahedral
What is the hybridization of each carbon atom?
$\boldsymbol{sp^3}$