QUESTION IMAGE
Question
question 1
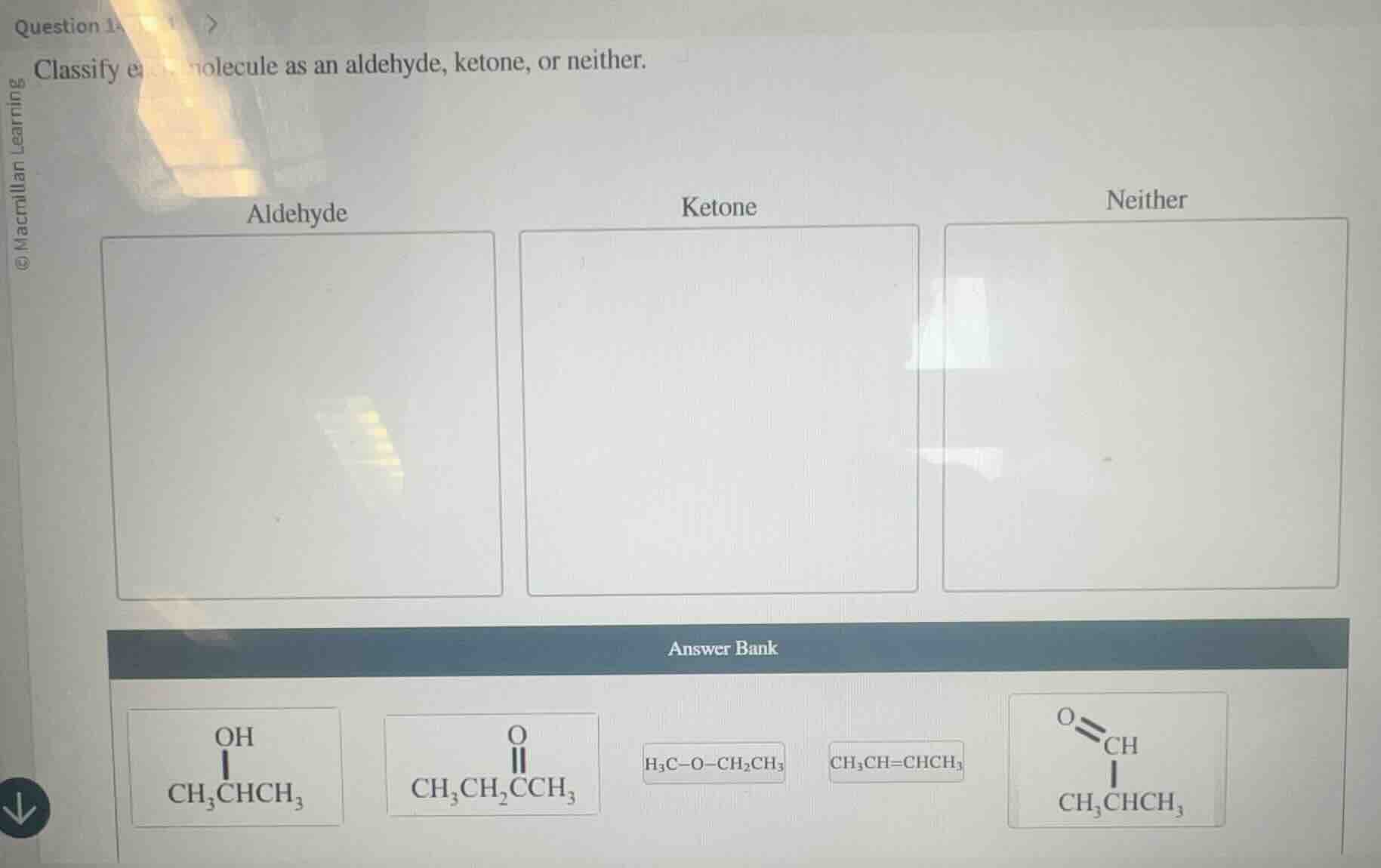
classify each molecule as an aldehyde, ketone, or neither.
aldehyde
ketone
neither
answer bank
\ce{ch3ch(oh)ch3}
\ce{ch3ch2coch3}
\ce{h3c-o-ch2ch3}
\ce{ch3ch=chch3}
\ce{ch3ch(ch3)coh}
Brief Explanations
- Aldehyde: Defined by a carbonyl group ($\text{-CHO}$) bonded to one alkyl group and one hydrogen. The molecule $\text{CH}_3\text{CHCH}_3\text{CHO}$ matches this structure.
- Ketone: Defined by a carbonyl group ($\text{C=O}$) bonded to two alkyl groups. The molecule $\text{CH}_3\text{CH}_2\text{C(O)CH}_3$ matches this structure.
- Neither: Molecules without aldehyde/ketone functional groups:
- $\text{CH}_3\text{CHCH}_3\text{OH}$ is an alcohol (has $\text{-OH}$ group).
- $\text{H}_3\text{C-O-CH}_2\text{CH}_3$ is an ether (has $\text{-O-}$ linkage).
- $\text{CH}_3\text{CH=CHCH}_3$ is an alkene (has $\text{C=C}$ double bond).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Aldehyde:
$\text{CH}_3\text{CHCH}_3\text{CHO}$ (or $\ce{CH3CH(CH3)CHO}$)
Ketone:
$\text{CH}_3\text{CH}_2\text{C(O)CH}_3$ (or $\ce{CH3CH2COCH3}$)
Neither:
$\text{CH}_3\text{CHCH}_3\text{OH}$ (or $\ce{CH3CH(CH3)OH}$), $\text{H}_3\text{C-O-CH}_2\text{CH}_3$ (or $\ce{CH3OCH2CH3}$), $\text{CH}_3\text{CH=CHCH}_3$