QUESTION IMAGE
Question
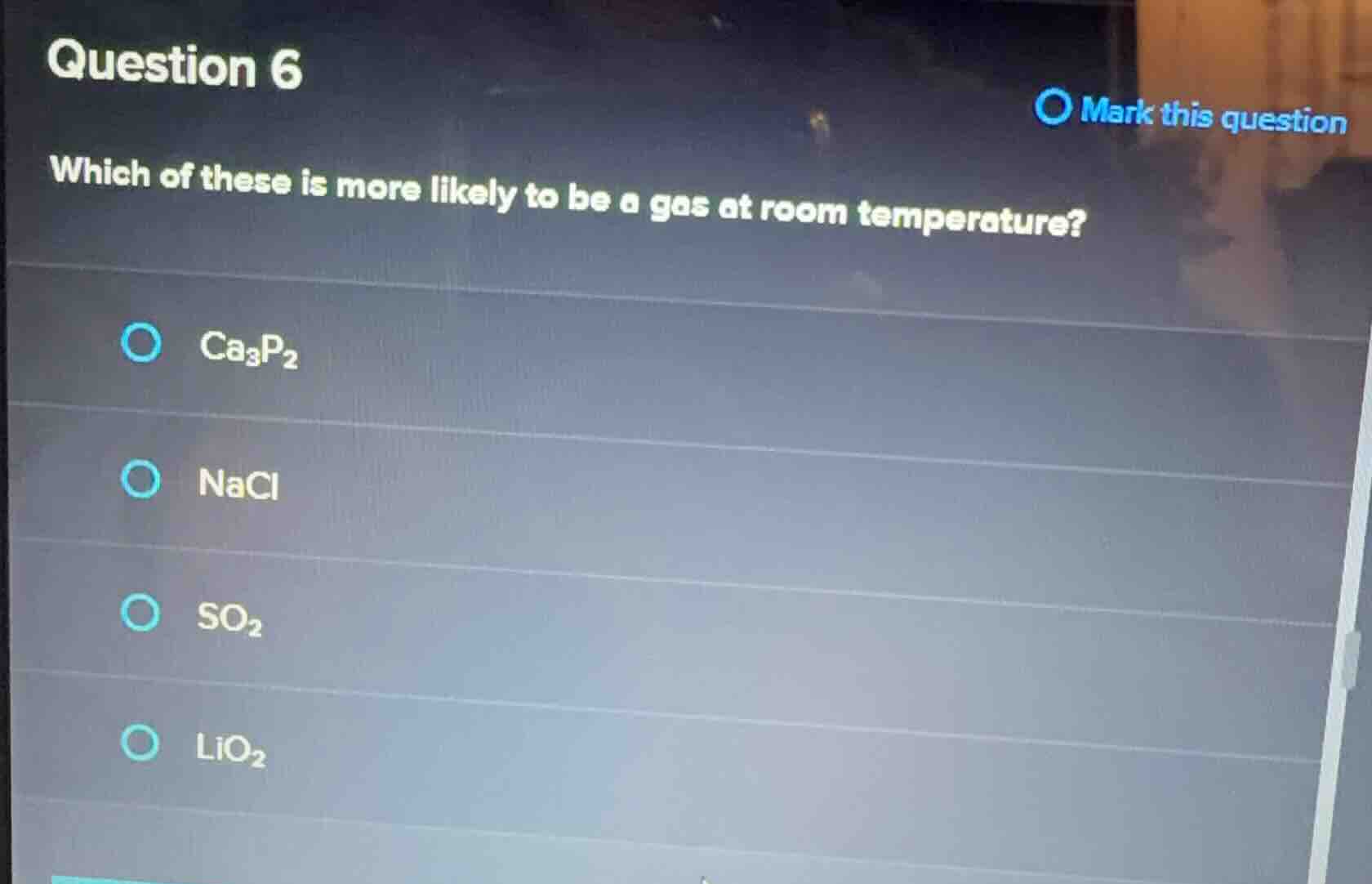
question 6
mark this question
which of these is more likely to be a gas at room temperature?
ca₃p₂
nacl
so₂
lio₂
Brief Explanations
Ionic compounds (like $\text{Ca}_3\text{P}_2$, $\text{NaCl}$, $\text{LiO}_2$) have strong ionic bonds, leading to high melting/boiling points, so they are solids at room temperature. $\text{SO}_2$ is a covalent molecular compound with weak intermolecular forces, which has a low boiling point ($-10^\circ\text{C}$), so it exists as a gas at room temperature.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. $\text{SO}_2$