QUESTION IMAGE
Question
question 3
5 pts
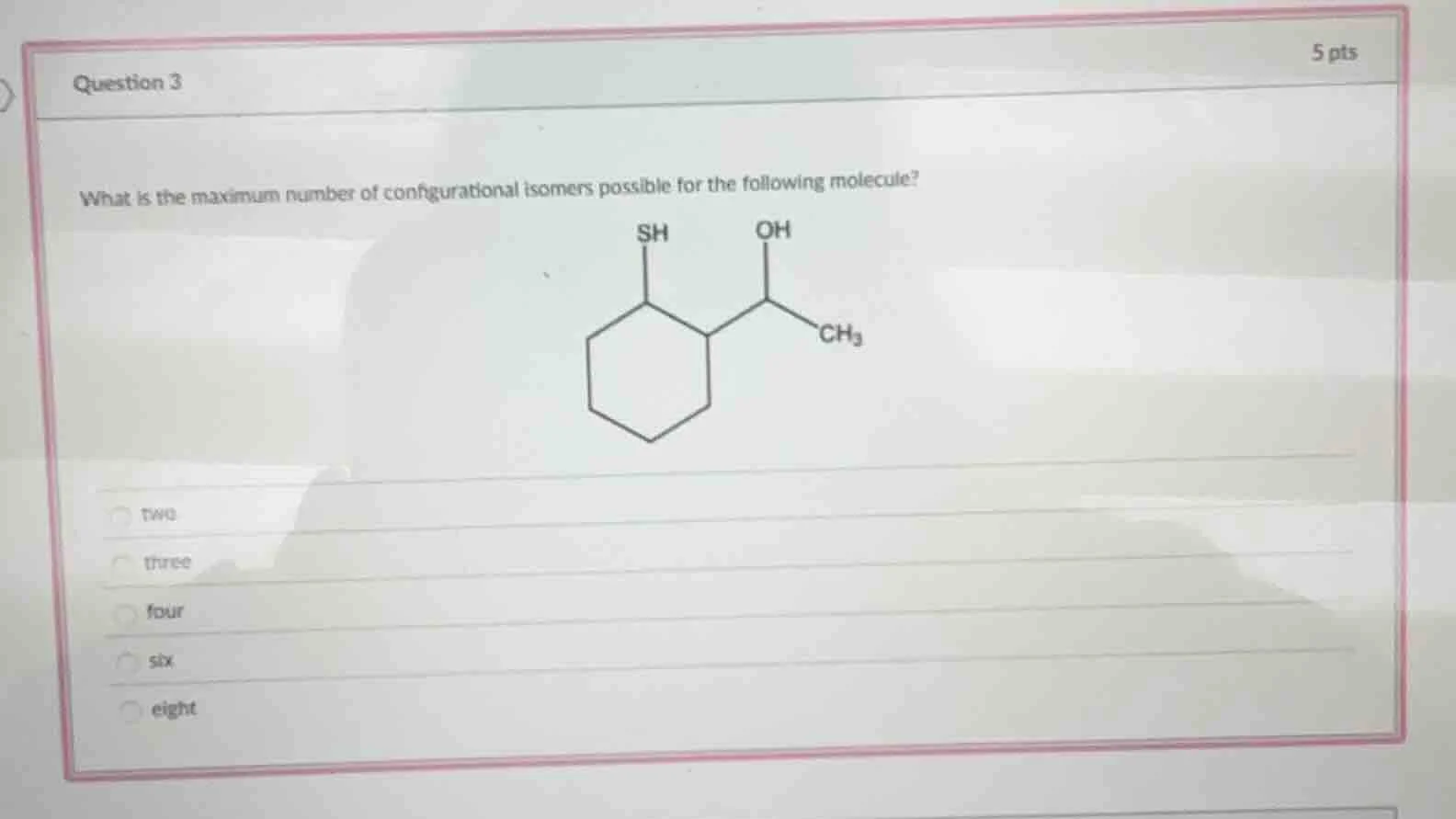
what is the maximum number of configurational isomers possible for the following molecule?
- two
- three
- four
- six
- eight
Step1: Identify chiral centers
The molecule has 2 distinct chiral centers: the cyclohexane carbon bonded to -SH, and the carbon bonded to -OH (both have 4 unique substituents).
Step2: Calculate max isomers
Use the formula $2^n$, where $n$ = number of chiral centers. Here, $n=2$.
$\boldsymbol{2^2 = 4}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
four