QUESTION IMAGE
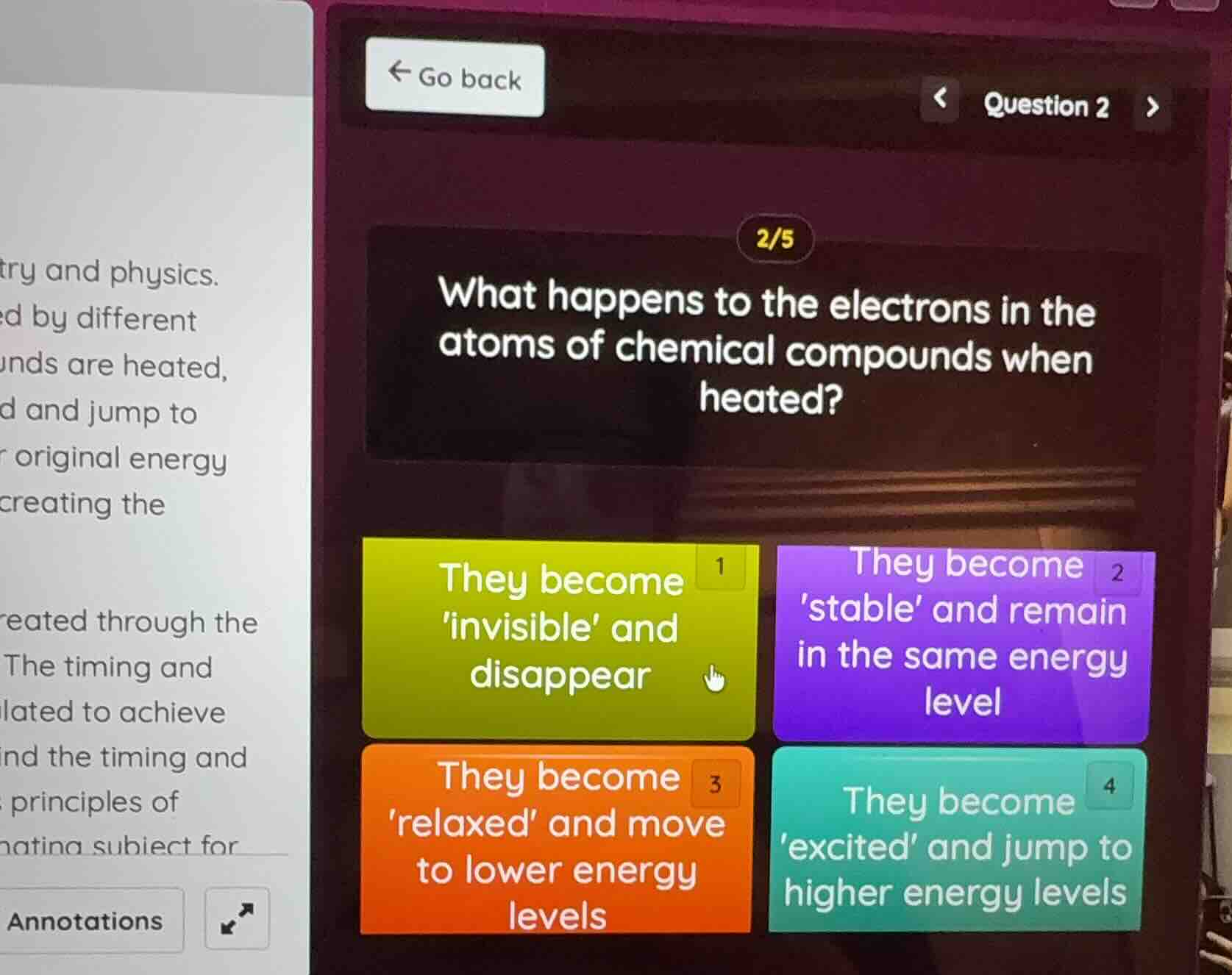
Question
question 2
what happens to the electrons in the atoms of chemical compounds when heated?
1 they become invisible and disappear
2 they become stable and remain in the same energy level
3 they become relaxed and move to lower energy levels
4 they become excited and jump to higher energy levels
When atoms in chemical compounds are heated, they absorb thermal energy. This energy is transferred to the electrons, causing them to gain energy and move from their normal, lower energy state to higher, less stable energy levels; this state is called being 'excited'.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- They become 'excited' and jump to higher energy levels