QUESTION IMAGE
Question
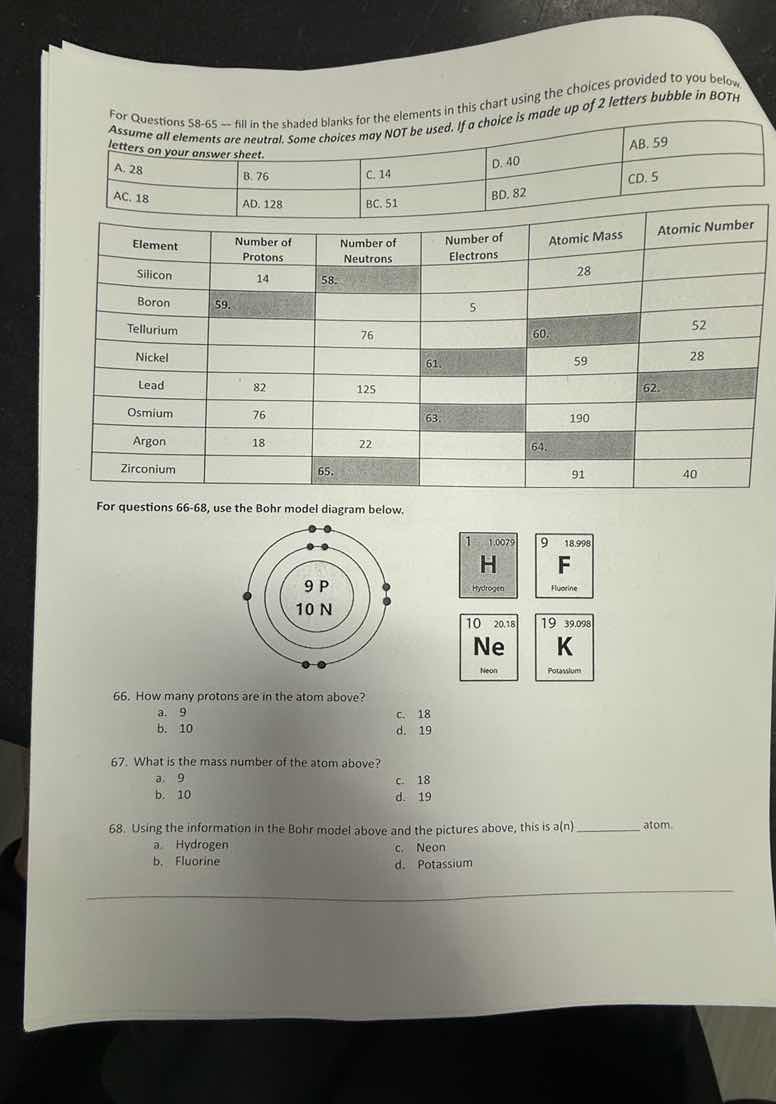
for questions 58-65 --- fill in the shaded blanks for the elements in this chart using the choices provided to you below. assume all elements are neutral. some choices may not be used. if a choice is made up of 2 letters bubble in both letters on your answer sheet. a. 28 b. 76 c. 14 d. 40 ab. 59 ac. 18 ad. 128 bc. 51 bd. 82 cd. 5 \
for questions 66-68, use the bohr model diagram below. bohr model diagram: nucleus with 9 p, 10 n, electron shells; and four element boxes: h, f, ne, k 66. how many protons are in the atom above? a. 9 b. 10 c. 18 d. 19 67. what is the mass number of the atom above? a. 9 b. 10 c. 18 d. 19 68. using the information in the bohr model above and the pictures above, this is a(n) \\_____ atom. a. hydrogen b. fluorine c. neon d. potassium
Question 66
In the Bohr model diagram, the number of protons is represented by the number labeled as "P" (protons). Here, it shows 9 P, so the number of protons is 9.
The mass number of an atom is the sum of the number of protons and neutrons. From the diagram, protons (P) = 9 and neutrons (N) = 10. So mass number = 9 + 10 = 19.
The atom has 9 protons (atomic number 9). Looking at the given elements, Fluorine (F) has an atomic number of 9 (from the box: 9 F). Hydrogen has atomic number 1, Neon 10, Potassium 19. So it's Fluorine.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
a. 9