QUESTION IMAGE
Question
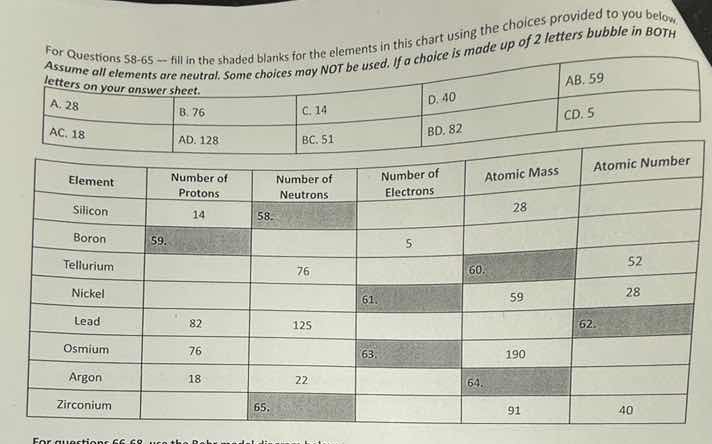
for questions s8 - 65 --- fill in the shaded blanks for the elements in this chart using the choices provided to you below
assume all elements are neutral. some choices may not be used. if a choice is made up of 2 letters bubble in both
letters on your answer sheet.
a. 28 b. 76 c. 14 d. 40 ab. 59
ac. 18 ad. 128 bc. 51 bd. 82 cd. 5
| element | number of protons | number of neutrons | number of electrons | atomic mass | atomic number |
|---|---|---|---|---|---|
| boron | 59. shaded | 5 | |||
| tellurium | 76 | 60. shaded | 52 | ||
| nickel | 61. shaded | 59 | 28 | ||
| lead | 82 | 125 | 62. shaded | ||
| osmium | 76 | 63. shaded | 190 | ||
| argon | 18 | 22 | 64. shaded | ||
| zirconium | 65. shaded | 91 | 40 |
for questions 66 - 68, use the bohr model...
To solve these fill - in - the - blank questions related to elements (atomic structure), we use the following concepts:
- For a neutral atom, the number of protons = the number of electrons = atomic number.
- Atomic mass = number of protons + number of neutrons.
Question 58 (Silicon - Number of Neutrons)
Step 1: Recall the formula for atomic mass
The formula for atomic mass is $Atomic\ Mass=Number\ of\ Protons + Number\ of\ Neutrons$. We know that for Silicon, the number of protons is 14 and the atomic mass is 28. Let the number of neutrons be $n$.
Step 2: Solve for the number of neutrons
From the formula $28 = 14 + n$, we can solve for $n$ by subtracting 14 from both sides of the equation. So, $n=28 - 14=14$. But wait, maybe we made a mistake. Wait, no, let's check again. Wait, the atomic mass of Silicon is actually 28.09, but in the context of this problem, we use the given values. Wait, the number of protons for Silicon is 14 (atomic number of Si is 14). The atomic mass is given as 28. So number of neutrons $=28 - 14 = 14$? But the options are A.28, B.76, C.14, D.40, AB.59, AC.18, AD.128, BC.51, BD.82, CD.5. So the answer for 58 is C.14.
Question 59 (Boron - Number of Protons/Atomic Number)
For a neutral atom, number of protons = atomic number = number of electrons. The number of electrons for Boron is 5 (from the table, the number of electrons is 5). So the number of protons (atomic number) should be 5. Looking at the options, CD is 5. So the answer for 59 is CD.5.
Question 60 (Tellurium - Atomic Mass)
Step 1: Recall the formula for atomic mass
$Atomic\ Mass = Number\ of\ Protons+Number\ of\ Neutrons$. The atomic number of Tellurium is 52, so the number of protons is 52. The number of neutrons is 76.
Step 2: Calculate atomic mass
$Atomic\ Mass=52 + 76=128$? Wait, no, wait the atomic number of Tellurium is 52, number of neutrons is 76. So $Atomic\ Mass = 52+76 = 128$, but the options: AD is 128. Wait, but let's check the table again. Wait, the atomic number of Tellurium is 52, number of neutrons is 76. So atomic mass $=52 + 76=128$, so the answer for 60 is AD.128.
Question 61 (Nickel - Number of Electrons)
Nickel has an atomic number of 28. For a neutral atom, number of electrons = atomic number. So the number of electrons is 28. The option A is 28. So the answer for 61 is A.28.
Question 62 (Lead - Atomic Number)
The number of protons in Lead is 82. For a neutral atom, atomic number = number of protons. So the atomic number is 82. The option BD is 82. So the answer for 62 is BD.82.
Question 63 (Osmium - Number of Electrons)
Osmium has 76 protons. For a neutral atom, number of electrons = number of protons. So the number of electrons is 76. The option B is 76. So the answer for 63 is B.76.
Question 64 (Argon - Atomic Mass)
Step 1: Recall the formula for atomic mass
$Atomic\ Mass=Number\ of\ Protons + Number\ of\ Neutrons$. The number of protons (atomic number) for Argon is 18, and the number of neutrons is 22.
Step 2: Calculate atomic mass
$Atomic\ Mass=18 + 22 = 40$. The option D is 40. So the answer for 64 is D.40.
Question 65 (Zirconium - Number of Neutrons)
Step 1: Recall the formula for atomic mass
$Atomic\ Mass=Number\ of\ Protons + Number\ of\ Neutrons$. The atomic number of Zirconium is 40 (so number of protons is 40), and the atomic mass is 91.
Step 2: Calculate number of neutrons
$Number\ of\ Neutrons=Atomic\ Mass - Number\ of\ Protons$. So $Number\ of\ Neutrons = 91-40=51$. The option BC is 51. So the answer for 65 is BC.51.
Final Answer…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To solve these fill - in - the - blank questions related to elements (atomic structure), we use the following concepts:
- For a neutral atom, the number of protons = the number of electrons = atomic number.
- Atomic mass = number of protons + number of neutrons.
Question 58 (Silicon - Number of Neutrons)
Step 1: Recall the formula for atomic mass
The formula for atomic mass is $Atomic\ Mass=Number\ of\ Protons + Number\ of\ Neutrons$. We know that for Silicon, the number of protons is 14 and the atomic mass is 28. Let the number of neutrons be $n$.
Step 2: Solve for the number of neutrons
From the formula $28 = 14 + n$, we can solve for $n$ by subtracting 14 from both sides of the equation. So, $n=28 - 14=14$. But wait, maybe we made a mistake. Wait, no, let's check again. Wait, the atomic mass of Silicon is actually 28.09, but in the context of this problem, we use the given values. Wait, the number of protons for Silicon is 14 (atomic number of Si is 14). The atomic mass is given as 28. So number of neutrons $=28 - 14 = 14$? But the options are A.28, B.76, C.14, D.40, AB.59, AC.18, AD.128, BC.51, BD.82, CD.5. So the answer for 58 is C.14.
Question 59 (Boron - Number of Protons/Atomic Number)
For a neutral atom, number of protons = atomic number = number of electrons. The number of electrons for Boron is 5 (from the table, the number of electrons is 5). So the number of protons (atomic number) should be 5. Looking at the options, CD is 5. So the answer for 59 is CD.5.
Question 60 (Tellurium - Atomic Mass)
Step 1: Recall the formula for atomic mass
$Atomic\ Mass = Number\ of\ Protons+Number\ of\ Neutrons$. The atomic number of Tellurium is 52, so the number of protons is 52. The number of neutrons is 76.
Step 2: Calculate atomic mass
$Atomic\ Mass=52 + 76=128$? Wait, no, wait the atomic number of Tellurium is 52, number of neutrons is 76. So $Atomic\ Mass = 52+76 = 128$, but the options: AD is 128. Wait, but let's check the table again. Wait, the atomic number of Tellurium is 52, number of neutrons is 76. So atomic mass $=52 + 76=128$, so the answer for 60 is AD.128.
Question 61 (Nickel - Number of Electrons)
Nickel has an atomic number of 28. For a neutral atom, number of electrons = atomic number. So the number of electrons is 28. The option A is 28. So the answer for 61 is A.28.
Question 62 (Lead - Atomic Number)
The number of protons in Lead is 82. For a neutral atom, atomic number = number of protons. So the atomic number is 82. The option BD is 82. So the answer for 62 is BD.82.
Question 63 (Osmium - Number of Electrons)
Osmium has 76 protons. For a neutral atom, number of electrons = number of protons. So the number of electrons is 76. The option B is 76. So the answer for 63 is B.76.
Question 64 (Argon - Atomic Mass)
Step 1: Recall the formula for atomic mass
$Atomic\ Mass=Number\ of\ Protons + Number\ of\ Neutrons$. The number of protons (atomic number) for Argon is 18, and the number of neutrons is 22.
Step 2: Calculate atomic mass
$Atomic\ Mass=18 + 22 = 40$. The option D is 40. So the answer for 64 is D.40.
Question 65 (Zirconium - Number of Neutrons)
Step 1: Recall the formula for atomic mass
$Atomic\ Mass=Number\ of\ Protons + Number\ of\ Neutrons$. The atomic number of Zirconium is 40 (so number of protons is 40), and the atomic mass is 91.
Step 2: Calculate number of neutrons
$Number\ of\ Neutrons=Atomic\ Mass - Number\ of\ Protons$. So $Number\ of\ Neutrons = 91-40=51$. The option BC is 51. So the answer for 65 is BC.51.
Final Answers:
- $\boldsymbol{C.14}$
- $\boldsymbol{CD.5}$
- $\boldsymbol{AD.128}$
- $\boldsymbol{A.28}$
- $\boldsymbol{BD.82}$
- $\boldsymbol{B.76}$
- $\boldsymbol{D.40}$
- $\boldsymbol{BC.51}$