QUESTION IMAGE
Question
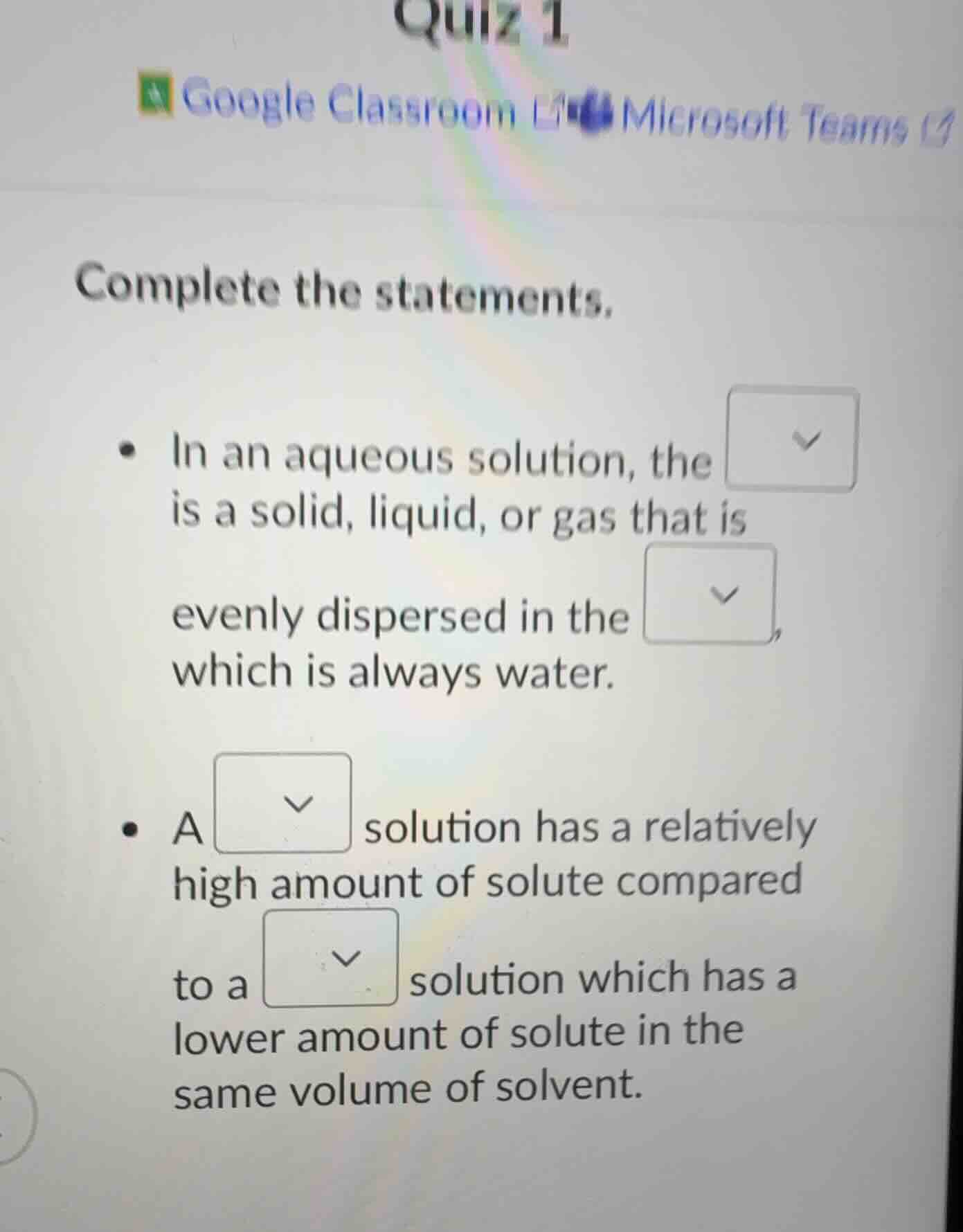
quiz 1 google classroom microsoft teams complete the statements. - in an aqueous solution, the is a solid, liquid, or gas that is evenly dispersed in the, which is always water. - a solution has a relatively high amount of solute compared to a solution which has a lower amount of solute in the same volume of solvent.
Brief Explanations
- For the first statement about aqueous solutions: In a solution, the solute is the substance (solid, liquid, gas) that gets dissolved, and the solvent is the substance doing the dissolving. In an aqueous solution, the solvent is water, so the first blank is "solute" and the second is "solvent".
- For the second statement about solution concentration: A concentrated solution has a relatively high amount of solute, while a dilute solution has a relatively low amount of solute (when comparing the same volume of solvent). So the first blank here is "concentrated" and the second is "dilute".
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- In an aqueous solution, the $\boldsymbol{\text{solute}}$ is a solid, liquid, or gas that is evenly dispersed in the $\boldsymbol{\text{solvent}}$, which is always water.
- A $\boldsymbol{\text{concentrated}}$ solution has a relatively high amount of solute compared to a $\boldsymbol{\text{dilute}}$ solution which has a lower amount of solute in the same volume of solvent.