QUESTION IMAGE
Question
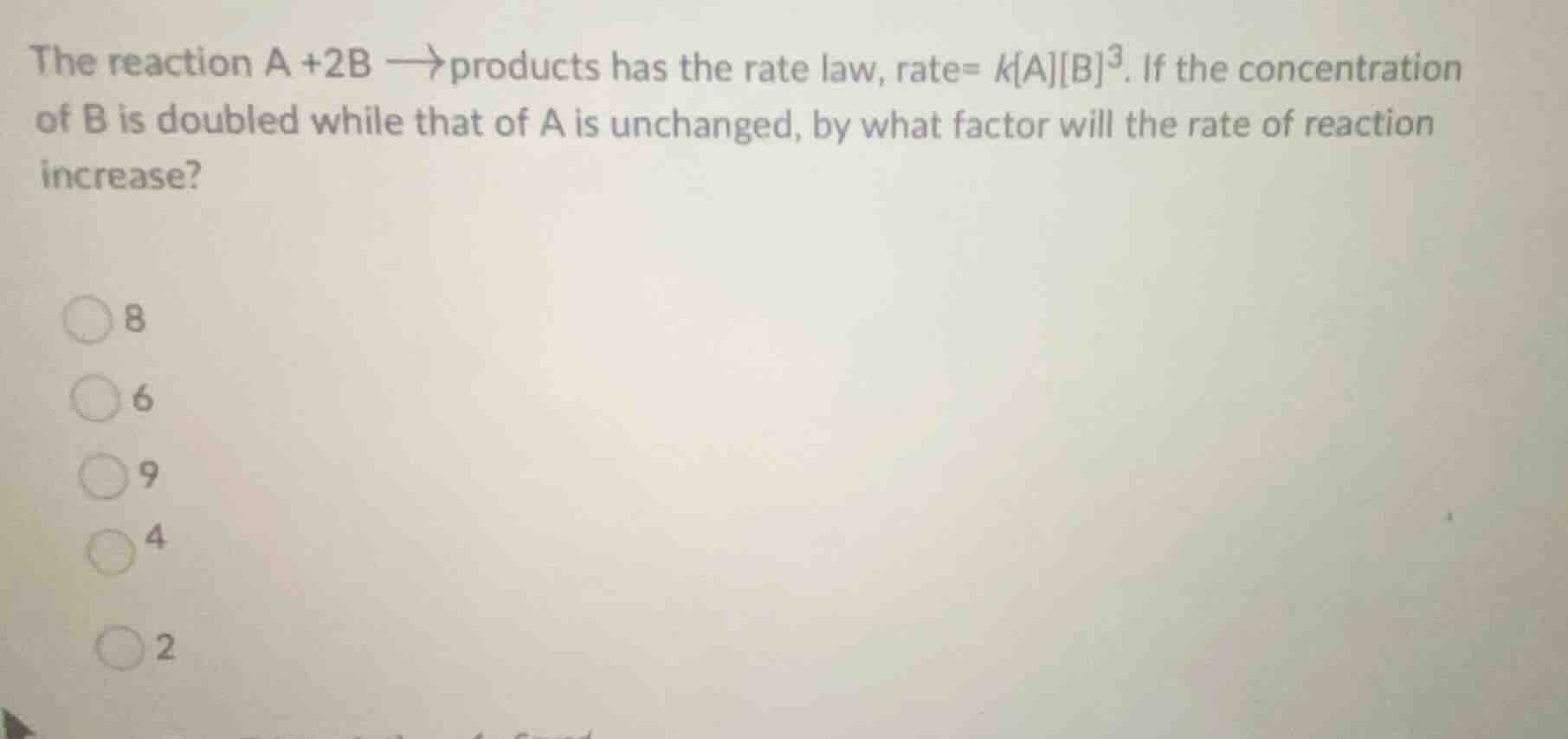
the reaction a +2b $
ightarrow$ products has the rate law, rate= $kab^3$. if the concentration of b is doubled while that of a is unchanged, by what factor will the rate of reaction increase?
8
6
9
4
2
Step1: Define initial reaction rate
$\text{rate}_1 = k[\text{A}][\text{B}]^3$
Step2: Substitute doubled [B]
$\text{rate}_2 = k[\text{A}](2[\text{B}])^3$
Step3: Simplify the new rate
$\text{rate}_2 = k[\text{A}] \times 8[\text{B}]^3 = 8k[\text{A}][\text{B}]^3$
Step4: Find rate ratio
$\frac{\text{rate}_2}{\text{rate}_1} = \frac{8k[\text{A}][\text{B}]^3}{k[\text{A}][\text{B}]^3} = 8$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
8