QUESTION IMAGE
Question
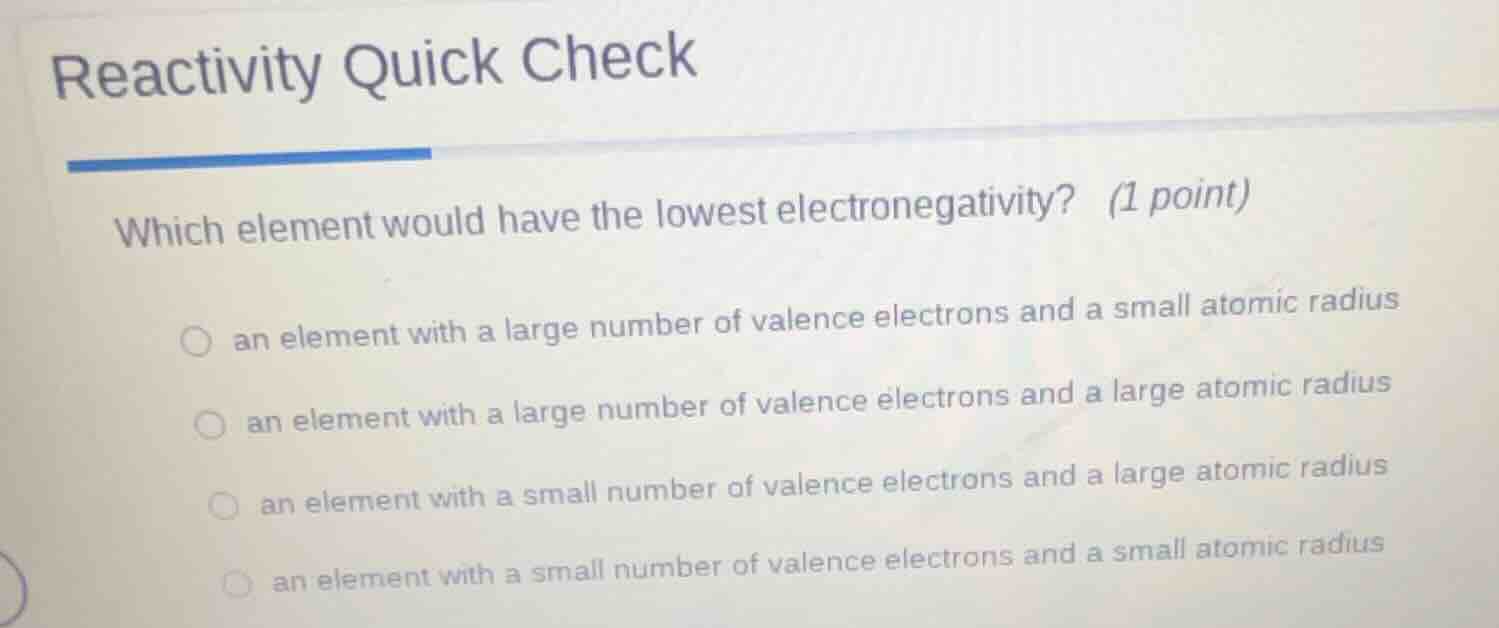
reactivity quick check
which element would have the lowest electronegativity? (1 point)
○ an element with a large number of valence electrons and a small atomic radius
○ an element with a large number of valence electrons and a large atomic radius
○ an element with a small number of valence electrons and a large atomic radius
○ an element with a small number of valence electrons and a small atomic radius
Electronegativity is the tendency of an atom to attract bonding electrons. It increases with fewer valence electrons (lower attraction for additional electrons, as atoms with few valence electrons tend to lose them) and larger atomic radius (the nucleus is farther from valence electrons, reducing its pull on external electrons).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
an element with a small number of valence electrons and a large atomic radius