QUESTION IMAGE
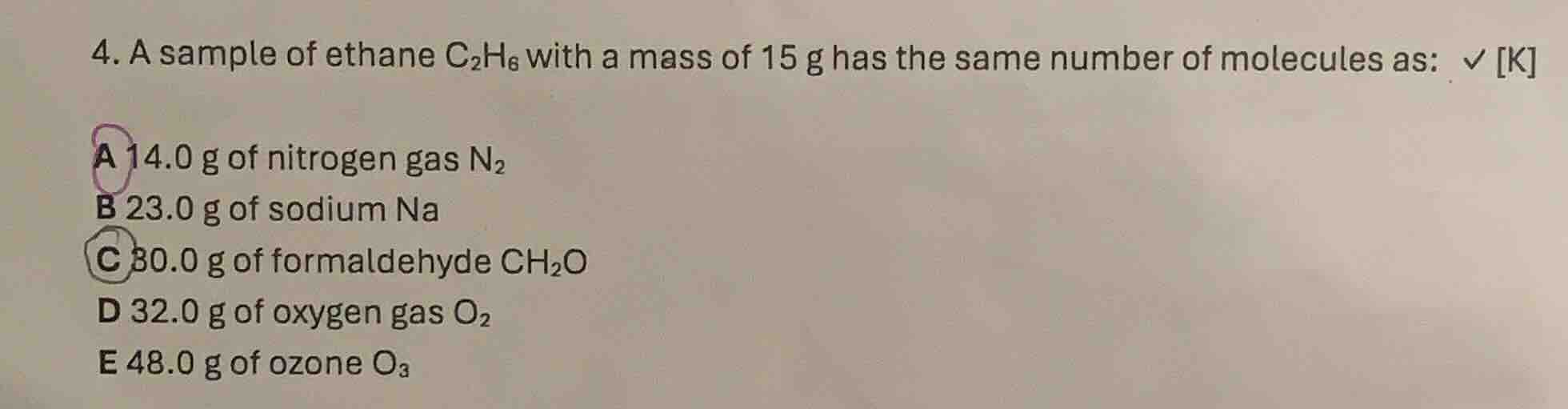
Question
- a sample of ethane c₂h₆ with a mass of 15 g has the same number of molecules as: k a 14.0 g of nitrogen gas n₂ b 23.0 g of sodium na c 30.0 g of formaldehyde ch₂o d 32.0 g of oxygen gas o₂ e 48.0 g of ozone o₃
Step1: Calculate moles of \( C_2H_6 \)
Molar mass of \( C_2H_6 \): \( 2\times12.01 + 6\times1.008 = 30.07 \, \text{g/mol} \)
Moles of \( C_2H_6 \): \( \frac{15 \, \text{g}}{30.07 \, \text{g/mol}} \approx 0.5 \, \text{mol} \)
Step2: Calculate moles for each option
- Option A (\( N_2 \)): Molar mass \( = 2\times14.01 = 28.02 \, \text{g/mol} \)
Moles: \( \frac{14.0 \, \text{g}}{28.02 \, \text{g/mol}} \approx 0.5 \, \text{mol} \)? Wait, \( 14/28 = 0.5 \)? Wait, \( 14.0 \, \text{g} / 28.02 \approx 0.5 \)? Wait, no: \( 14.0 / 28.02 \approx 0.5 \)? Wait, \( 28.02 \times 0.5 = 14.01 \), so yes, ~0.5 mol? Wait, but let's check others.
- Option B (\( \text{Na} \)): Molar mass \( = 22.99 \, \text{g/mol} \)
Moles: \( \frac{23.0 \, \text{g}}{22.99 \, \text{g/mol}} \approx 1.0 \, \text{mol} \)
- Option C (\( \text{CH}_2\text{O} \)): Molar mass \( = 12.01 + 2\times1.008 + 16.00 = 30.03 \, \text{g/mol} \)
Moles: \( \frac{30.0 \, \text{g}}{30.03 \, \text{g/mol}} \approx 1.0 \, \text{mol} \)
- Option D (\( \text{O}_2 \)): Molar mass \( = 2\times16.00 = 32.00 \, \text{g/mol} \)
Moles: \( \frac{32.0 \, \text{g}}{32.00 \, \text{g/mol}} = 1.0 \, \text{mol} \)
- Option E (\( \text{O}_3 \)): Molar mass \( = 3\times16.00 = 48.00 \, \text{g/mol} \)
Moles: \( \frac{48.0 \, \text{g}}{48.00 \, \text{g/mol}} = 1.0 \, \text{mol} \)
Wait, correction: For \( C_2H_6 \), \( 15 \, \text{g} / 30.07 \approx 0.5 \, \text{mol} \). For \( N_2 \), \( 14.0 \, \text{g} / 28.02 \approx 0.5 \, \text{mol} \)? Wait, \( 14.0 / 28.02 \approx 0.5 \) (since \( 28 \times 0.5 = 14 \)). Wait, but let's recheck \( C_2H_6 \): \( 15 \, \text{g} / 30.07 \approx 0.5 \, \text{mol} \). Then \( N_2 \): \( 14.0 \, \text{g} / 28.02 \approx 0.5 \, \text{mol} \). But wait, the original problem’s marked answer is A? Wait, no, wait: Wait, \( C_2H_6 \) molar mass is ~30, so 15g is 0.5mol. \( N_2 \) molar mass 28, 14g is 0.5mol. But wait, let's check the options again. Wait, maybe I made a mistake. Wait, the question is "same number of molecules", which means same moles (since \( \text{molecules} = n \times N_A \)). So we need moles = 0.5 mol.
Wait, \( C_2H_6 \): \( 15 / 30 = 0.5 \, \text{mol} \) (approx).
- A: \( 14 / 28 = 0.5 \, \text{mol} \)
- B: \( 23 / 23 = 1 \, \text{mol} \) (Na molar mass ~23)
- C: \( 30 / 30 = 1 \, \text{mol} \) (CH₂O molar mass ~30)
- D: \( 32 / 32 = 1 \, \text{mol} \) (O₂ molar mass 32)
- E: \( 48 / 48 = 1 \, \text{mol} \) (O₃ molar mass 48)
So A has 0.5 mol, same as \( C_2H_6 \) (0.5 mol). Wait, but the original image has A and C marked? Wait, no, the user’s image shows A is circled and C is also circled? Wait, maybe a typo. Wait, no, let's recalculate:
Wait, \( C_2H_6 \) molar mass: \( 2\times12 + 6\times1 = 30 \, \text{g/mol} \). So 15g is \( 15/30 = 0.5 \, \text{mol} \).
- A: \( N_2 \) molar mass 28, 14g is \( 14/28 = 0.5 \, \text{mol} \).
- B: Na molar mass 23, 23g is 1mol.
- C: CH₂O molar mass 30, 30g is 1mol.
- D: O₂ molar mass 32, 32g is 1mol.
- E: O₃ molar mass 48, 48g is 1mol.
So A has 0.5 mol, same as \( C_2H_6 \) (0.5 mol). So the correct answer is A.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. 14.0 g of nitrogen gas \( \text{N}_2 \)