QUESTION IMAGE
Question
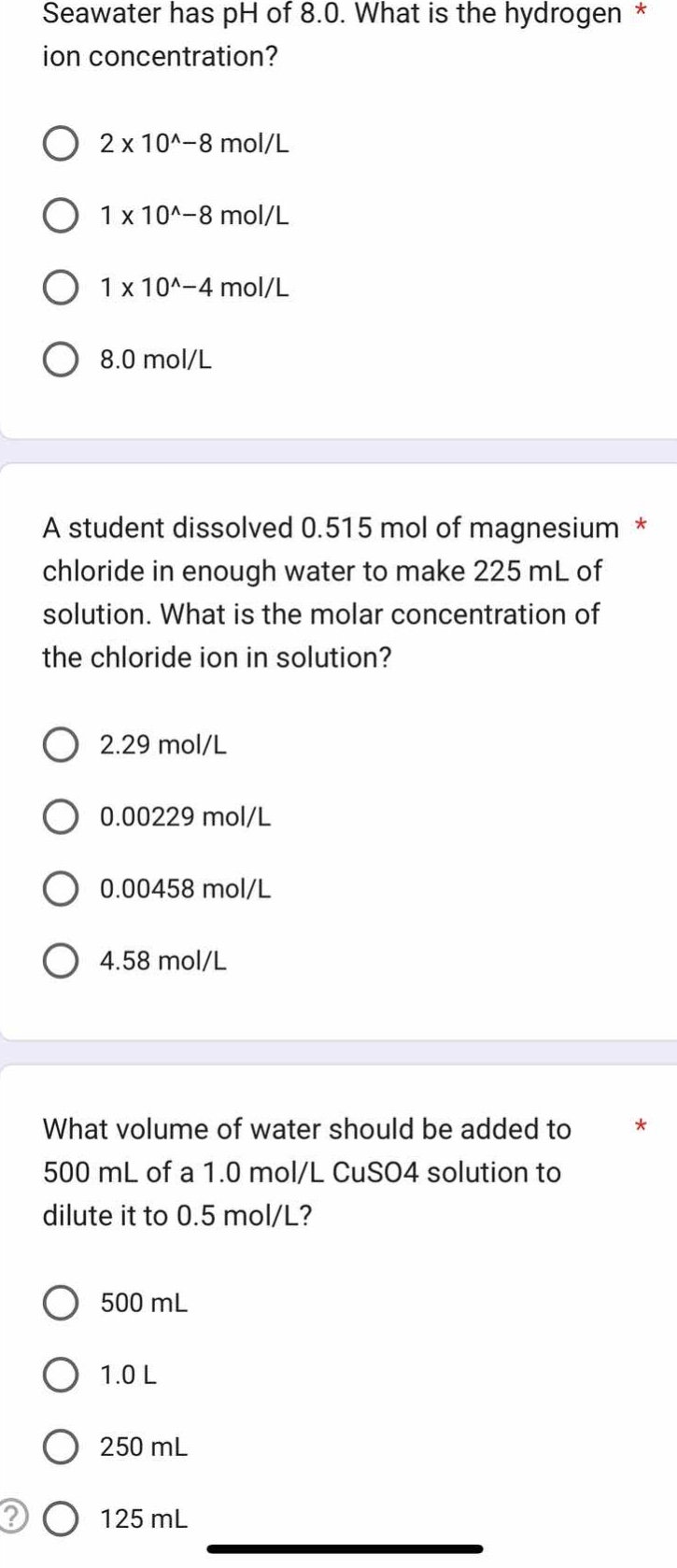
seawater has ph of 8.0. what is the hydrogen ion concentration?
2 x 10^-8 mol/l
1 x 10^-8 mol/l
1 x 10^-4 mol/l
8.0 mol/l
a student dissolved 0.515 mol of magnesium chloride in enough water to make 225 ml of solution. what is the molar concentration of the chloride ion in solution?
2.29 mol/l
0.00229 mol/l
0.00458 mol/l
4.58 mol/l
what volume of water should be added to 500 ml of a 1.0 mol/l cuso4 solution to dilute it to 0.5 mol/l?
500 ml
1.0 l
250 ml
125 ml
---
Problem 1:
Step1: Recall pH definition
The formula for pH is $\text{pH} = -\log_{10}[\text{H}^+]$, where $[\text{H}^+]$ is hydrogen ion concentration.
Step2: Rearrange for $[\text{H}^+]$
$[\text{H}^+] = 10^{-\text{pH}}$
Step3: Substitute pH=8.0
$[\text{H}^+] = 10^{-8.0} = 1 \times 10^{-8}\ \text{mol/L}$
---
Problem 2:
Step1: Convert volume to liters
$225\ \text{mL} = \frac{225}{1000} = 0.225\ \text{L}$
Step2: Find $\text{MgCl}_2$ molarity
Molarity of $\text{MgCl}_2 = \frac{\text{moles}}{\text{volume}} = \frac{0.515}{0.225} \approx 2.2889\ \text{mol/L}$
Step3: Relate to $\text{Cl}^-$ concentration
1 mole $\text{MgCl}_2$ gives 2 moles $\text{Cl}^-$, so $[\text{Cl}^-] = 2 \times 2.2889 \approx 4.58\ \text{mol/L}$
---
Problem 3:
Step1: Use dilution formula
Dilution formula: $C_1V_1 = C_2V_2$, where $C$ = concentration, $V$ = total volume.
Step2: Solve for final volume $V_2$
$V_2 = \frac{C_1V_1}{C_2} = \frac{1.0\ \text{mol/L} \times 500\ \text{mL}}{0.5\ \text{mol/L}} = 1000\ \text{mL}$
Step3: Calculate water to add
Water added = $V_2 - V_1 = 1000\ \text{mL} - 500\ \text{mL} = 500\ \text{mL}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- B. $1 \times 10^{-8}\ \text{mol/L}$
- D. $4.58\ \text{mol/L}$
- A. $500\ \text{mL}$