QUESTION IMAGE
Question
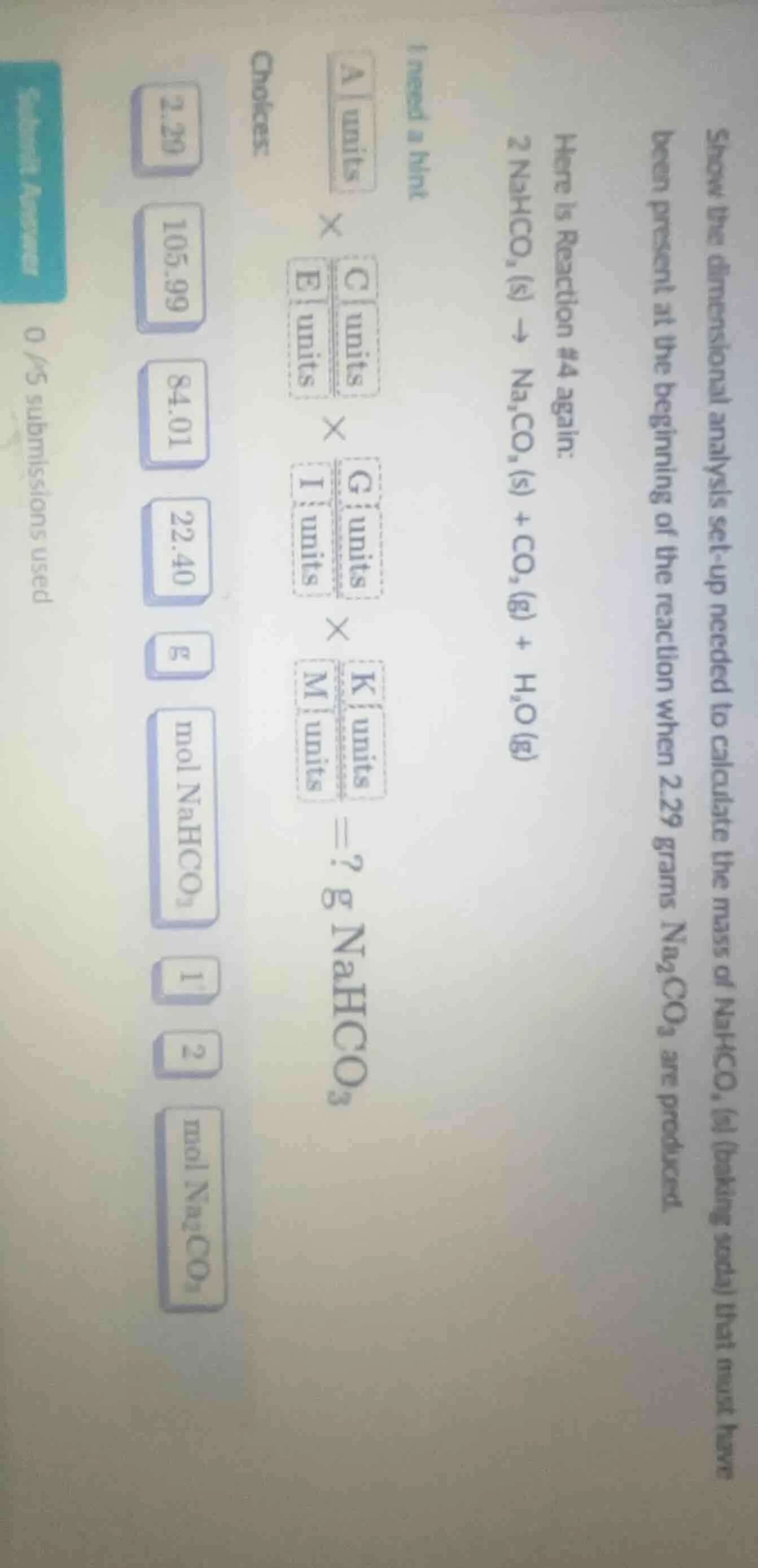
show the dimensional analysis set-up needed to calculate the mass of nahco₃ (s) (baking soda) that must have been present at the beginning of the reaction when 2.29 grams na₂co₃ are produced.
here is reaction #4 again:
2 nahco₃ (s) → na₂co₃ (s) + co₂ (g) + h₂o (g)
i need a hint
$\boldsymbol{a units} \times \boldsymbol{c units} \times \frac{\boldsymbol{g units}}{\boldsymbol{i units}} \times \frac{\boldsymbol{k units}}{\boldsymbol{m units}} = ? \text{g} \text{nahco}_3$
choces:
2.29, 105.99, 84.01, 22.40, g, mol nahco₃, 1, 2, mol na₂co₃
0 /5 submissions used
Step1: Start with given mass
$2.29\ \text{g}\ \text{Na}_2\text{CO}_3$
Step2: Convert to moles of $\text{Na}_2\text{CO}_3$
$\frac{1\ \text{mol}\ \text{Na}_2\text{CO}_3}{105.99\ \text{g}\ \text{Na}_2\text{CO}_3}$
Step3: Mole ratio from reaction
$\frac{2\ \text{mol}\ \text{NaHCO}_3}{1\ \text{mol}\ \text{Na}_2\text{CO}_3}$
Step4: Convert to grams of $\text{NaHCO}_3$
$\frac{84.01\ \text{g}\ \text{NaHCO}_3}{1\ \text{mol}\ \text{NaHCO}_3}$
Step5: Combine all factors
$2.29\ \text{g}\ \text{Na}_2\text{CO}_3 \times \frac{1\ \text{mol}\ \text{Na}_2\text{CO}_3}{105.99\ \text{g}\ \text{Na}_2\text{CO}_3} \times \frac{2\ \text{mol}\ \text{NaHCO}_3}{1\ \text{mol}\ \text{Na}_2\text{CO}_3} \times \frac{84.01\ \text{g}\ \text{NaHCO}_3}{1\ \text{mol}\ \text{NaHCO}_3}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The complete dimensional analysis setup is:
$2.29\ \text{g}\ \text{Na}_2\text{CO}_3 \times \frac{1\ \text{mol}\ \text{Na}_2\text{CO}_3}{105.99\ \text{g}\ \text{Na}_2\text{CO}_3} \times \frac{2\ \text{mol}\ \text{NaHCO}_3}{1\ \text{mol}\ \text{Na}_2\text{CO}_3} \times \frac{84.01\ \text{g}\ \text{NaHCO}_3}{1\ \text{mol}\ \text{NaHCO}_3} = 3.60\ \text{g}\ \text{NaHCO}_3$
Filled into the template:
A: $2.29\ \text{g}\ \text{Na}_2\text{CO}_3$
C: $\frac{1\ \text{mol}\ \text{Na}_2\text{CO}_3}{105.99\ \text{g}\ \text{Na}_2\text{CO}_3}$
E: $\frac{2\ \text{mol}\ \text{NaHCO}_3}{1\ \text{mol}\ \text{Na}_2\text{CO}_3}$
G: $\frac{84.01\ \text{g}\ \text{NaHCO}_3}{1\ \text{mol}\ \text{NaHCO}_3}$
Final calculated mass: $3.60\ \text{g}\ \text{NaHCO}_3$