QUESTION IMAGE
Question
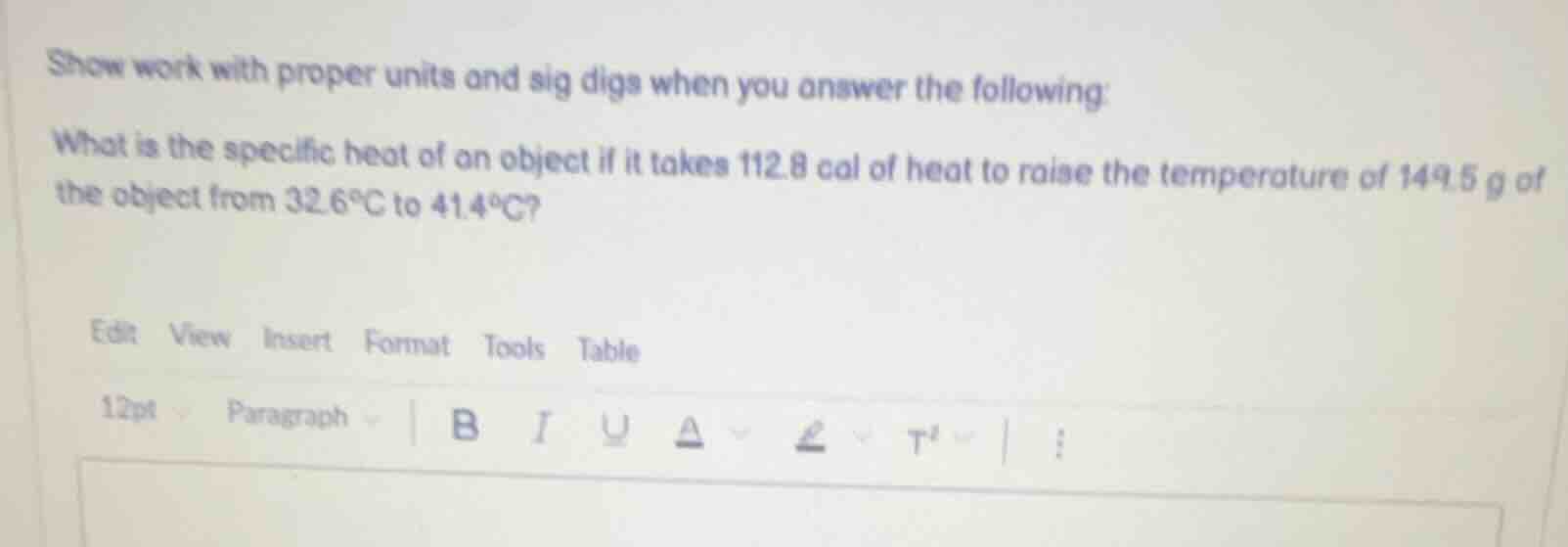
show work with proper units and sig digs when you answer the following: what is the specific heat of an object if it takes 112.8 cal of heat to raise the temperature of 149.5 g of the object from 32.6°c to 41.4°c?
Step1: Define given values
$Q = 112.8\ \text{cal}$, $m = 149.5\ \text{g}$, $T_i = 32.6^\circ\text{C}$, $T_f = 41.4^\circ\text{C}$
Step2: Calculate temperature change
$\Delta T = T_f - T_i = 41.4^\circ\text{C} - 32.6^\circ\text{C} = 8.8^\circ\text{C}$
Step3: Rearrange heat formula for $c$
Use $Q = mc\Delta T$, solve for $c$: $c = \frac{Q}{m\Delta T}$
Step4: Substitute values and solve
$c = \frac{112.8\ \text{cal}}{149.5\ \text{g} \times 8.8^\circ\text{C}} = \frac{112.8}{1315.6}\ \frac{\text{cal}}{\text{g} \cdot ^\circ\text{C}} \approx 0.0858\ \frac{\text{cal}}{\text{g} \cdot ^\circ\text{C}}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$0.0858\ \frac{\text{cal}}{\text{g} \cdot ^\circ\text{C}}$