QUESTION IMAGE
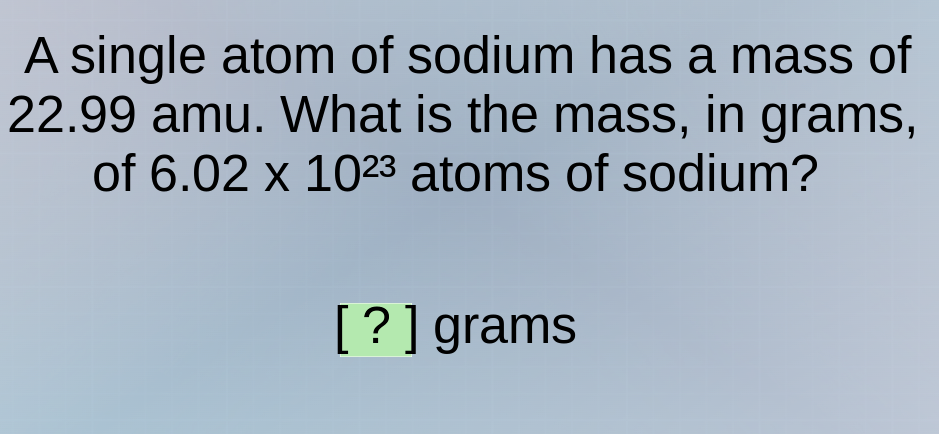
Question
a single atom of sodium has a mass of 22.99 amu. what is the mass, in grams, of 6.02 x 10²³ atoms of sodium? ? grams
Step1: Recall Avogadro's number and molar mass concept
Avogadro's number is \(6.02\times10^{23}\) atoms/mol, and the molar mass of an element (in grams per mole) is numerically equal to its atomic mass in amu. So, for sodium, the atomic mass is 22.99 amu, which means its molar mass is 22.99 g/mol. This means that \(6.02\times10^{23}\) atoms of sodium (which is 1 mole of sodium atoms) will have a mass equal to its molar mass.
Step2: Calculate the mass
Since 1 mole of sodium atoms (\(6.02\times10^{23}\) atoms) has a molar mass of 22.99 g/mol, the mass of \(6.02\times10^{23}\) atoms of sodium is equal to its molar mass. So the mass is 22.99 grams.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
22.99