QUESTION IMAGE
Question
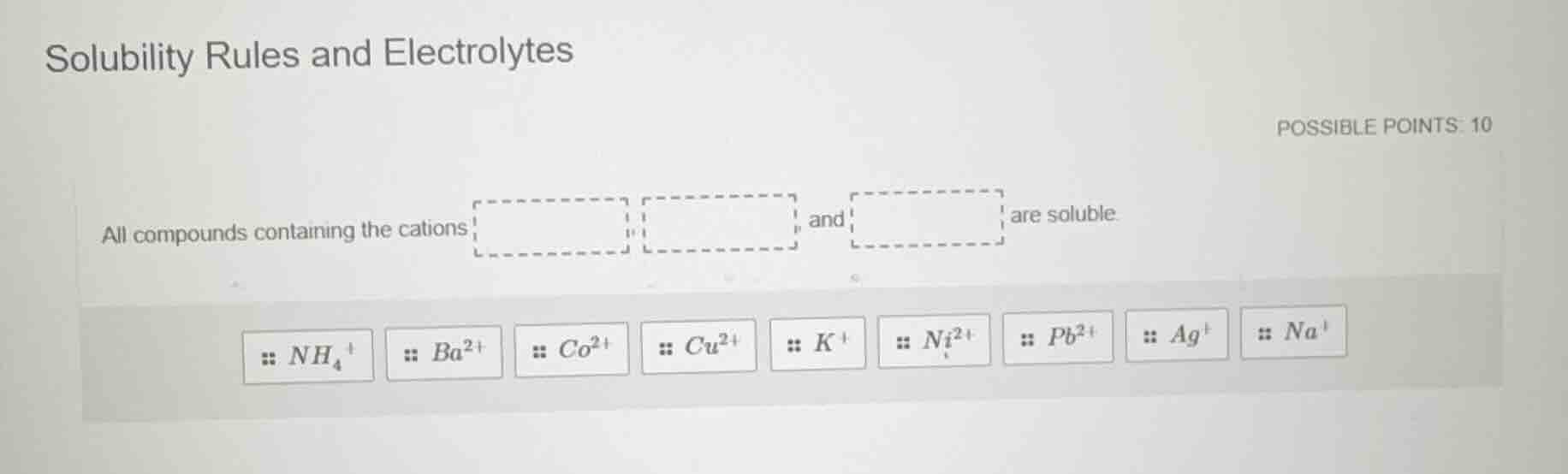
solubility rules and electrolytes
possible points: 10
all compounds containing the cations and are soluble
$nh_4^+$ $ba^{2+}$ $co^{2+}$ $cu^{2+}$ $k^+$ $ni^{2+}$ $pb^{2+}$ $ag^+$ $na^+$
Brief Explanations
Solubility rules state that all compounds containing group 1 metal cations (like sodium, potassium) and ammonium cations are universally soluble in water, with no exceptions. The other cations listed form insoluble compounds with some anions.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$NH_4^+$, $K^+$, $Na^+$