QUESTION IMAGE
Question
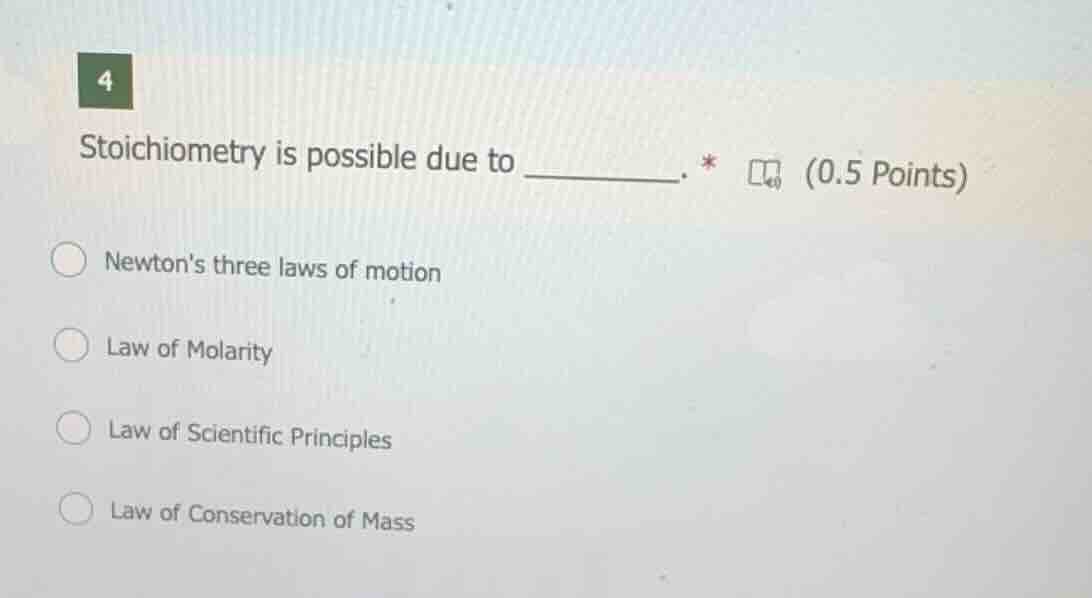
4
stoichiometry is possible due to ______. * (0.5 points)
newtons three laws of motion
law of molarity
law of scientific principles
law of conservation of mass
Stoichiometry deals with the quantitative relationships in chemical reactions. The Law of Conservation of Mass (matter is neither created nor destroyed in a chemical reaction) is the basis for stoichiometric calculations, as it allows us to balance reactions and relate amounts of reactants and products. Newton’s laws relate to motion, the “Law of Molarity” is not a fundamental law, and “Law of Scientific Principles” is not a specific law related to stoichiometry.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
D. Law of Conservation of Mass