QUESTION IMAGE
Question
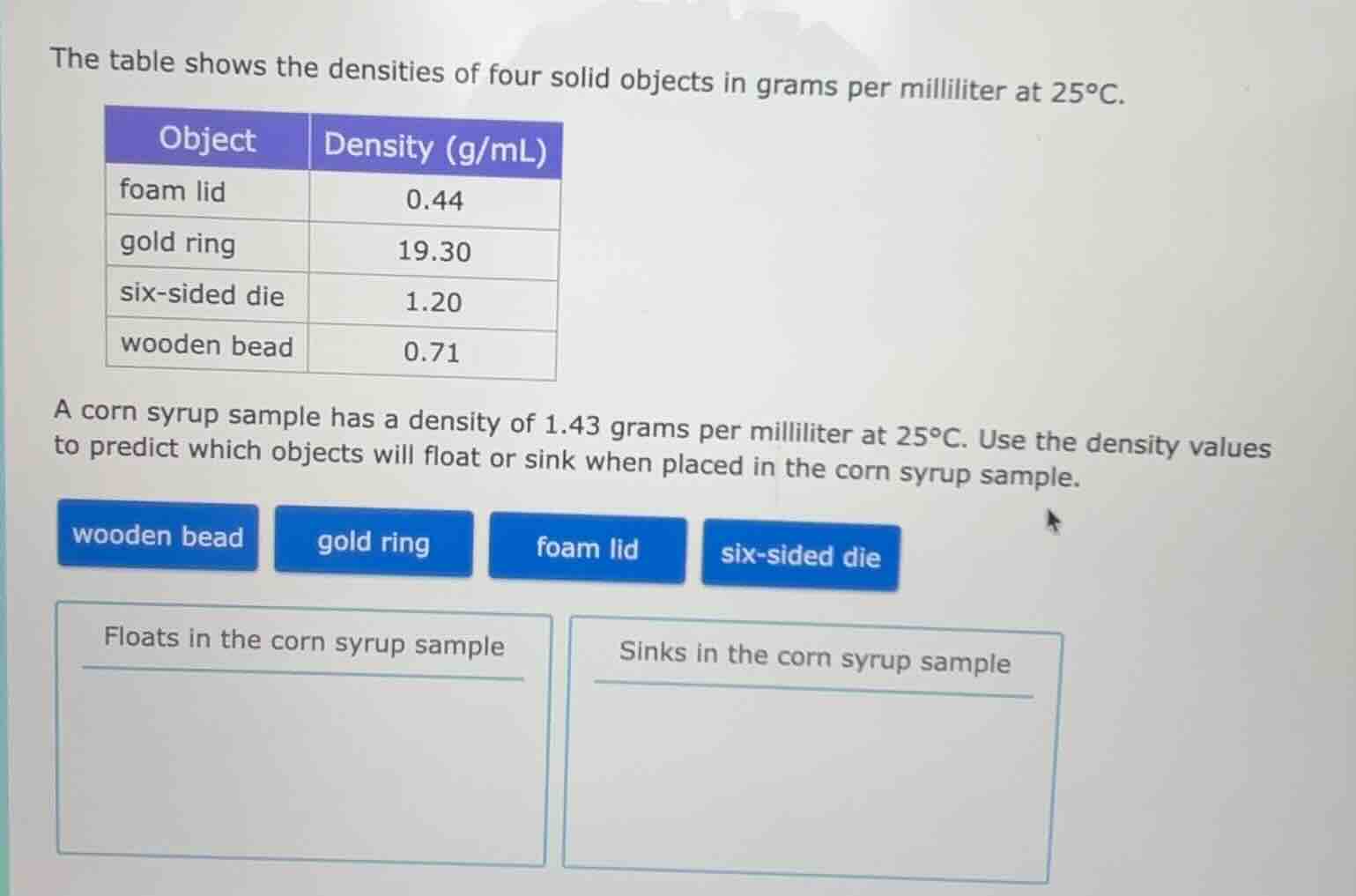
the table shows the densities of four solid objects in grams per milliliter at 25°c.
object | density (g/ml)
foam lid | 0.44
gold ring | 19.30
six - sided die | 1.20
wooden bead | 0.71
a corn syrup sample has a density of 1.43 grams per milliliter at 25°c. use the density values to predict which objects will float or sink when placed in the corn syrup sample.
wooden bead gold ring foam lid six - sided die
floats in the corn syrup sample
sinks in the corn syrup sample
Step1: Recall float/sink rule
An object floats if its density is less than the fluid's density, sinks if greater.
Step2: Compare each object's density to 1.43 g/mL
- Foam lid: $0.44 < 1.43$
- Wooden bead: $0.71 < 1.43$
- Six-sided die: $1.20 < 1.43$
- Gold ring: $19.30 > 1.43$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Floats in the corn syrup sample:
foam lid, wooden bead, six-sided die
Sinks in the corn syrup sample:
gold ring