QUESTION IMAGE
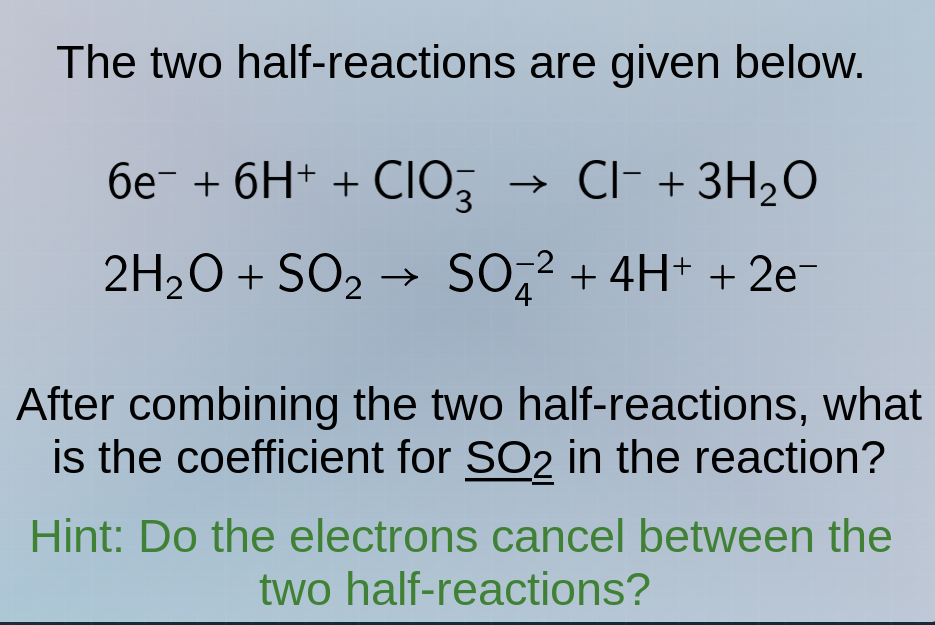
Question
the two half - reactions are given below.
$6e^- + 6h^+ + clo_3^- \
ightarrow cl^- + 3h_2o$
$2h_2o + so_2 \
ightarrow so_4^{ - 2} + 4h^+ + 2e^-$
after combining the two half - reactions, what is the coefficient for $so_2$ in the reaction?
hint: do the electrons cancel between the two half - reactions?
Step1: Balance electrons in half-reactions
First half-reaction (reduction) has 6 electrons gained. Second half-reaction (oxidation) has 2 electrons lost. To balance electrons, multiply the oxidation half-reaction by 3 (since \(6\div2 = 3\)).
Oxidation half-reaction (after multiplying by 3):
\(3\times(2H_2O + SO_2
ightarrow SO_4^{2-} + 4H^+ + 2e^-)\)
\(6H_2O + 3SO_2
ightarrow 3SO_4^{2-} + 12H^+ + 6e^-\)
Step2: Combine the two half-reactions
Reduction half-reaction: \(6e^- + 6H^+ + ClO_3^-
ightarrow Cl^- + 3H_2O\)
Now, add the two half-reactions (electrons cancel out):
\(6H_2O + 3SO_2 + 6e^- + 6H^+ + ClO_3^-
ightarrow 3SO_4^{2-} + 12H^+ + 6e^- + Cl^- + 3H_2O\)
Simplify (cancel \(6e^-\), subtract \(6H^+\) from \(12H^+\), subtract \(3H_2O\) from \(6H_2O\)):
\(3H_2O + 3SO_2 + ClO_3^-
ightarrow 3SO_4^{2-} + 6H^+ + Cl^-\)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
3