QUESTION IMAGE
Question
types of reactions
quiz complete
1 2 3 4 5 6 7
time remaining
46:27
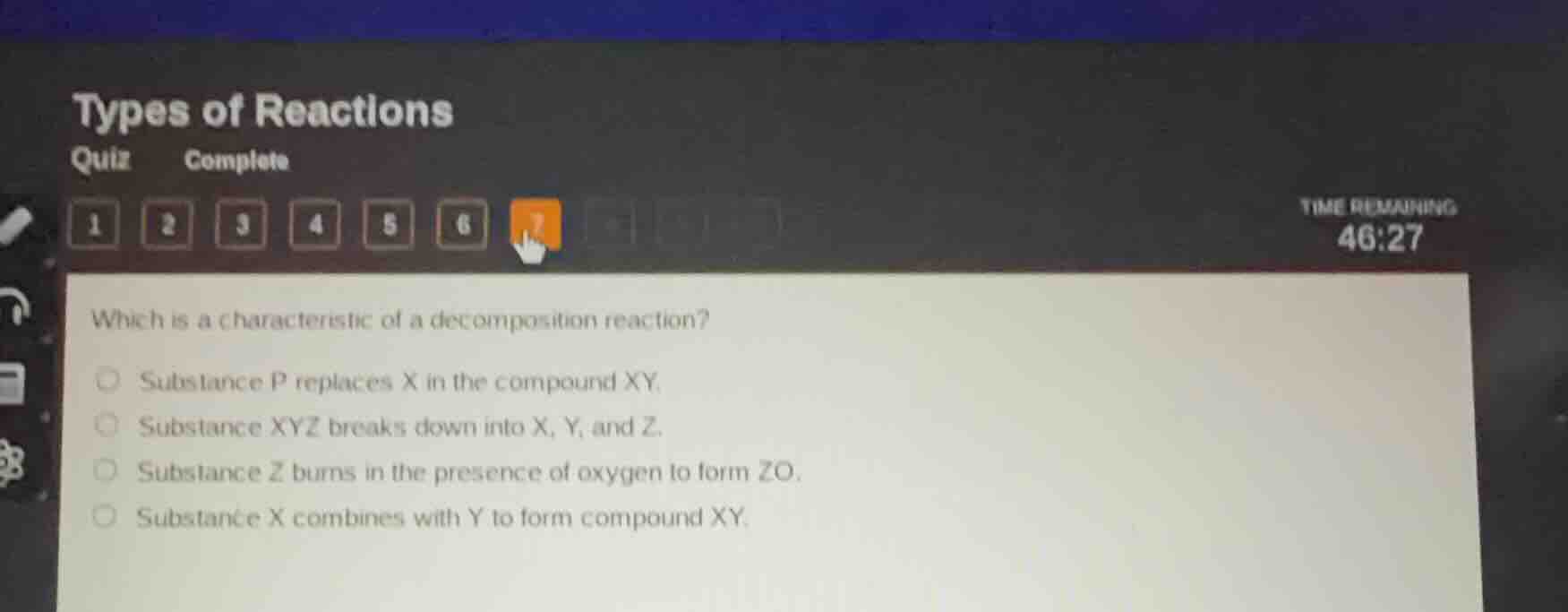
which is a characteristic of a decomposition reaction?
substance p replaces x in the compound xy.
substance xyz breaks down into x, y, and z.
substance z burns in the presence of oxygen to form zo.
substance x combines with y to form compound xy.
Brief Explanations
A decomposition reaction is defined as a chemical reaction where a single compound breaks down into two or more simpler substances. Let's analyze each option:
- "Substance P replaces X in the compound XY" describes a single displacement reaction, not decomposition.
- "Substance XYZ breaks down into X, Y, and Z" matches the definition of a decomposition reaction, as one compound splits into multiple simpler substances.
- "Substance Z burns in the presence of oxygen to form ZO" describes a combustion reaction, not decomposition.
- "Substance X combines with Y to form compound XY" describes a synthesis (combination) reaction, not decomposition.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Substance XYZ breaks down into X, Y, and Z.