QUESTION IMAGE
Question
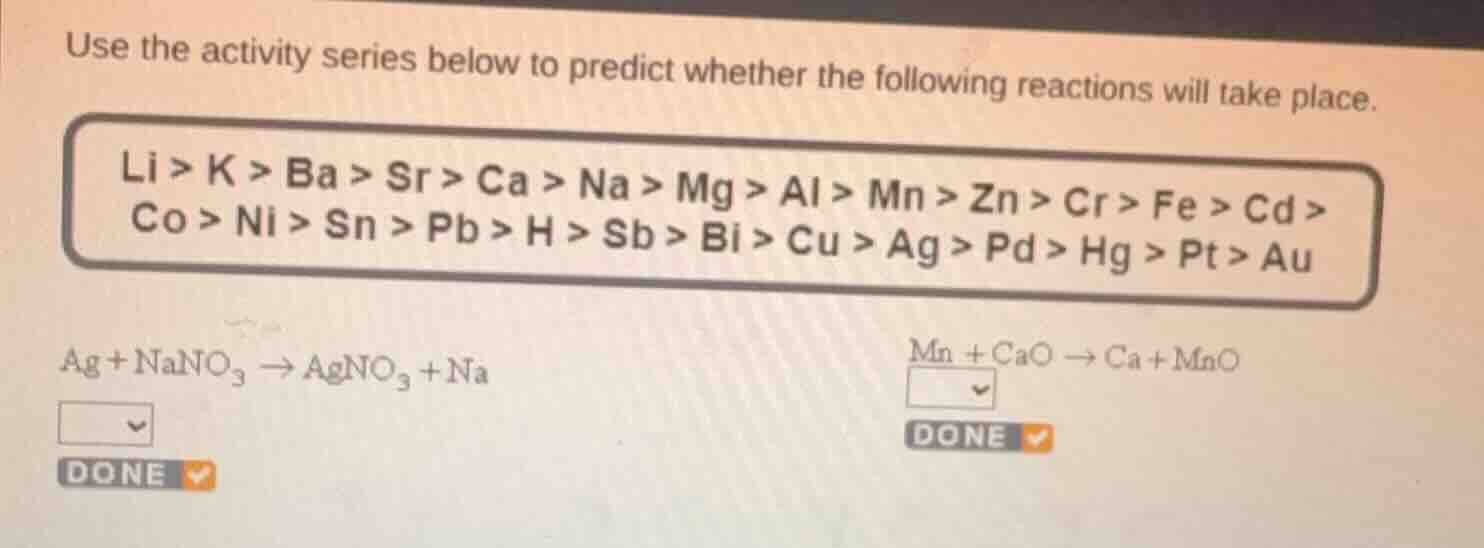
use the activity series below to predict whether the following reactions will take place.
li > k > ba > sr > ca > na > mg > al > mn > zn > cr > fe > cd >
co > ni > sn > pb > h > sb > bi > cu > ag > pd > hg > pt > au
ag + nano₃ → agno₃ + na
mn + cao → ca + mno
For the reaction \( \boldsymbol{\text{Ag} + \text{NaNO}_3
ightarrow \text{AgNO}_3 + \text{Na}} \)
Step1: Identify metal activities
From the activity series, \( \text{Na} > \text{Ag} \) (since \( \text{Na} \) is to the left of \( \text{Ag} \) in the series \( \text{Li} > \text{K} > \dots > \text{Na} > \dots > \text{Ag} > \dots \)).
Step2: Apply displacement rule
A metal can displace another metal from its compound only if it is more active (higher in the activity series) than the metal in the compound. Here, \( \text{Ag} \) is less active than \( \text{Na} \), so \( \text{Ag} \) cannot displace \( \text{Na} \) from \( \text{NaNO}_3 \). Thus, the reaction does not occur.
For the reaction \( \boldsymbol{\text{Mn} + \text{CaO}
ightarrow \text{Ca} + \text{MnO}} \)
Step1: Identify metal activities
From the activity series, \( \text{Ca} > \text{Mn} \) (since \( \text{Ca} \) is to the left of \( \text{Mn} \) in the series \( \text{Li} > \dots > \text{Ca} > \dots > \text{Mn} > \dots \)).
Step2: Apply displacement rule
A metal can displace another metal from its compound only if it is more active (higher in the activity series) than the metal in the compound. Here, \( \text{Mn} \) is less active than \( \text{Ca} \), so \( \text{Mn} \) cannot displace \( \text{Ca} \) from \( \text{CaO} \). Thus, the reaction does not occur.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
s:
- For \( \text{Ag} + \text{NaNO}_3
ightarrow \text{AgNO}_3 + \text{Na} \): The reaction does not take place.
- For \( \text{Mn} + \text{CaO}
ightarrow \text{Ca} + \text{MnO} \): The reaction does not take place.