QUESTION IMAGE
Question
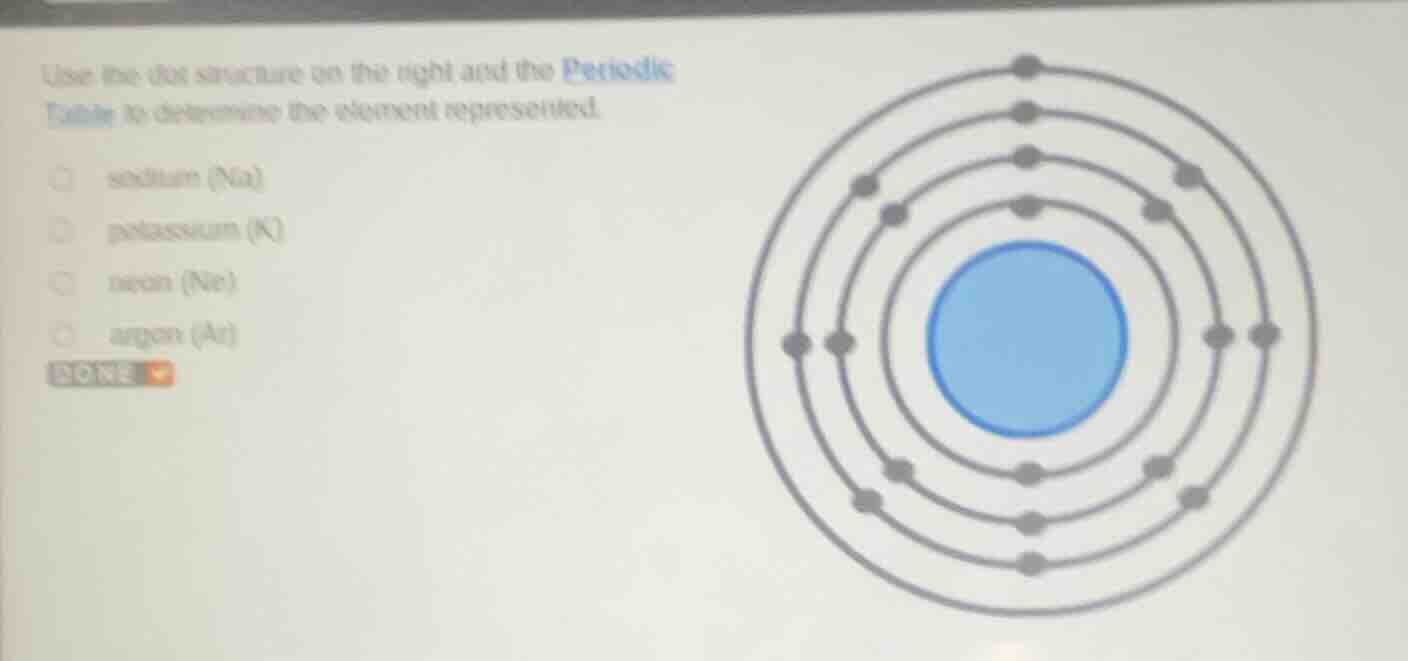
use the dot structure on the right and the periodic table to determine the element represented. sodium (na) potassium (k) neon (ne) argon (ar) done
To determine the element, we analyze the electron dot structure (electron configuration). Let's count the electrons in each shell:
- First shell (closest to nucleus): 2 electrons.
- Second shell: 8 electrons.
- Third shell: 8 electrons.
- Fourth shell: 1 electron.
The total number of electrons (which equals the atomic number for a neutral atom) is \(2 + 8 + 8 + 1 = 19\). The element with atomic number 19 is potassium (K). Sodium (Na) has atomic number 11, neon (Ne) has 10, and argon (Ar) has 18. So the correct answer is potassium (K).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. potassium (K)