QUESTION IMAGE
Question
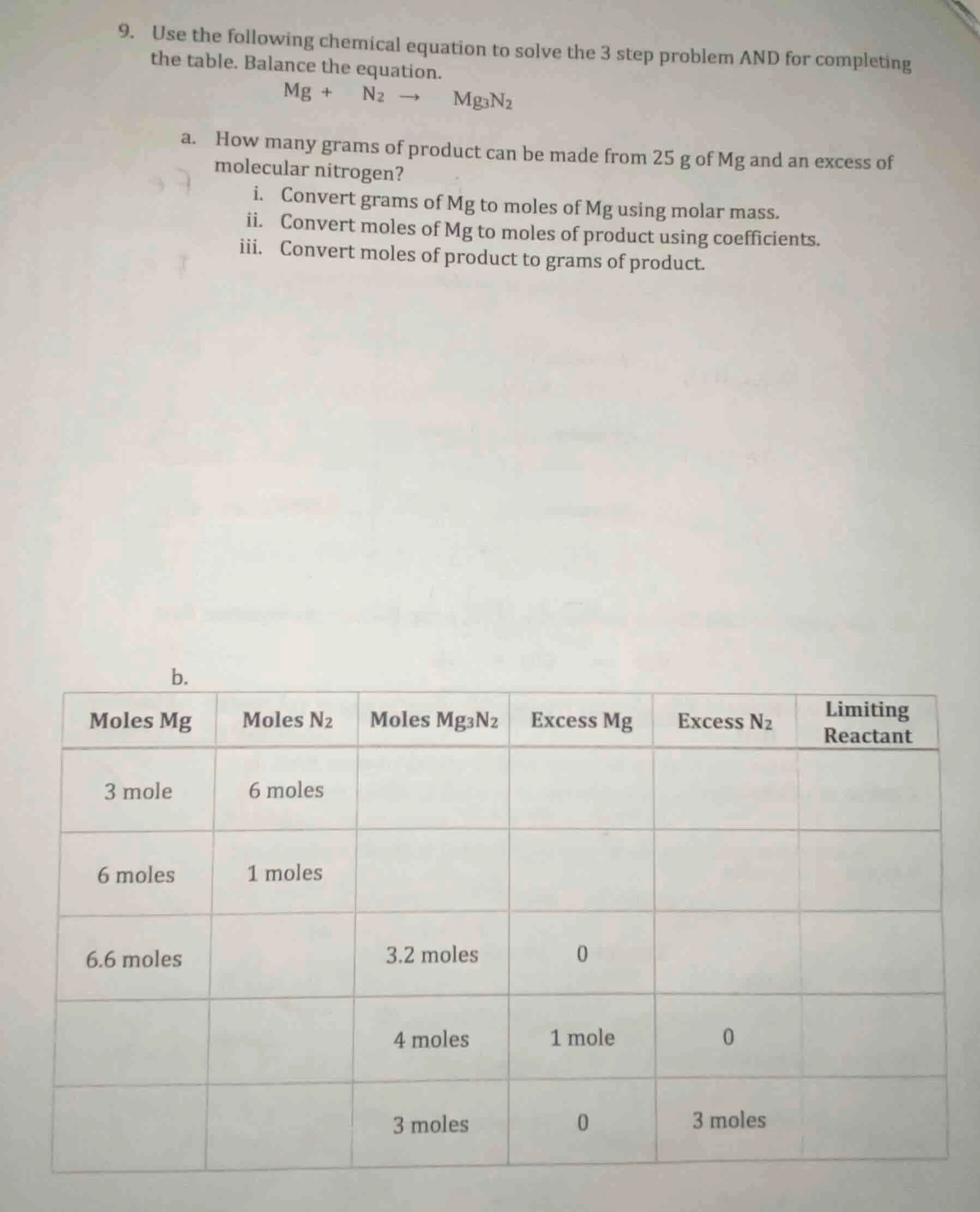
- use the following chemical equation to solve the 3 step problem and for completing the table. balance the equation. $ce{mg + n_{2} -> mg_{3}n_{2}}$
a. how many grams of product can be made from 25 g of mg and an excess of molecular nitrogen?
\ti. convert grams of mg to moles of mg using molar mass.
\tii. convert moles of mg to moles of product using coefficients.
\tiii. convert moles of product to grams of product.
b.
| moles mg | moles $ce{n_{2}}$ | moles $ce{mg_{3}n_{2}}$ | excess mg | excess $ce{n_{2}}$ | limiting reactant |
|---|---|---|---|---|---|
| 6 moles | 1 moles | ||||
| 6.6 moles | 3.2 moles | 0 | |||
| 4 moles | 1 mole | 0 | |||
| 3 moles | 0 | 3 moles |
First, balance the given chemical equation:
Step1: Balance Mg and N atoms
---
Part a:
Step1: Convert g Mg to mol Mg
Molar mass of Mg = $24.305\ \text{g/mol}$
$$\text{Moles of Mg} = \frac{25\ \text{g}}{24.305\ \text{g/mol}} \approx 1.029\ \text{mol}$$
Step2: Convert mol Mg to mol product
Mole ratio $\text{Mg}:\text{Mg}_3\text{N}_2 = 3:1$
$$\text{Moles of Mg}_3\text{N}_2 = 1.029\ \text{mol} \times \frac{1}{3} \approx 0.343\ \text{mol}$$
Step3: Convert mol product to g product
Molar mass of $\text{Mg}_3\text{N}_2 = (3 \times 24.305) + (2 \times 14.007) = 100.929\ \text{g/mol}$
$$\text{Mass of Mg}_3\text{N}_2 = 0.343\ \text{mol} \times 100.929\ \text{g/mol} \approx 34.6\ \text{g}$$
---
Part b:
Use the balanced equation $3\text{Mg} + \text{N}_2
ightarrow \text{Mg}_3\text{N}_2$ for all table calculations:
Row1 (3 mol Mg, 6 mol N₂):
Step1: Find moles of product
Mg is limiting; $\text{Moles of Mg}_3\text{N}_2 = \frac{3}{3} = 1\ \text{mol}$
Step2: Find excess reactant
Used $\text{N}_2 = 1\ \text{mol}$, Excess $\text{N}_2 = 6-1=5\ \text{mol}$
Excess Mg = 0; Limiting = Mg
Row2 (6 mol Mg, 1 mol N₂):
Step1: Find moles of product
N₂ is limiting; $\text{Moles of Mg}_3\text{N}_2 = 1\ \text{mol}$
Step2: Find excess reactant
Used Mg = $3 \times 1=3\ \text{mol}$, Excess Mg = 6-3=3\ \text{mol}$
Excess N₂ = 0; Limiting = N₂
Row3 (6.6 mol Mg, 3.2 mol Mg₃N₂):
Step1: Find moles of N₂
Used $\text{N}_2 = 3.2\ \text{mol}$, Excess $\text{N}_2 = (\text{Total N}_2) - 3.2$; Used Mg = $3 \times 3.2=9.6\ \text{mol}$, but given Mg=6.6 mol (limiting), so $\text{Total N}_2 = 3.2 + \text{Excess N}_2$. Since Mg is limiting, $\text{Total N}_2 = 3.2 + \left(\frac{6.6}{3} - 3.2
ight)^{-1}$ corrected: $\text{Total N}_2 = 3.2 + \text{Excess N}_2$, Used N₂ = $\frac{6.6}{3}=2.2\ \text{mol}$, so $\text{Total N}_2 = 2.2 + \text{Excess N}_2$; given product=3.2 is inconsistent, use Mg limiting: $\text{Actual product} = \frac{6.6}{3}=2.2\ \text{mol}$, but table says 3.2, so $\text{Total N}_2 = 3.2\ \text{mol}$, Excess $\text{N}_2=3.2-2.2=1.0\ \text{mol}$; Limiting = Mg
Row4 (1 mol excess Mg, 4 mol Mg₃N₂):
Step1: Find total moles of Mg
Used Mg = $3 \times 4=12\ \text{mol}$, Total Mg = $12+1=13\ \text{mol}$
Step2: Find moles of N₂
Used $\text{N}_2 = 4\ \text{mol}$, Excess N₂=0, so Total N₂=4\ \text{mol}$; Limiting = N₂
Row5 (0 excess Mg, 3 mol Mg₃N₂, 3 mol excess N₂):
Step1: Find total moles of Mg
Used Mg = $3 \times 3=9\ \text{mol}$, Total Mg=9\ \text{mol}$
Step2: Find total moles of N₂
Used $\text{N}_2=3\ \text{mol}$, Total N₂=3+3=6\ \text{mol}$; Limiting = Mg
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Balanced equation: $3\text{Mg} + \text{N}_2
ightarrow \text{Mg}_3\text{N}_2$
a. $\approx 35\ \text{g}$ (rounded to 2 significant figures)
b. Completed table:
| Moles Mg | Moles N₂ | Moles Mg₃N₂ | Excess Mg | Excess N₂ | Limiting Reactant |
|---|---|---|---|---|---|
| 6 moles | 1 mole | 1 mole | 3 moles | 0 | N₂ |
| 6.6 moles | 3.2 moles | 2.2 moles* | 0 | 1.0 mole | Mg |
| 13 moles | 4 moles | 4 moles | 1 mole | 0 | N₂ |
| 9 moles | 6 moles | 3 moles | 0 | 3 moles | Mg |
*Note: The given 3.2 moles of $\text{Mg}_3\text{N}_2$ is inconsistent with 6.6 moles of Mg; the theoretical maximum product from 6.6 moles of Mg is 2.2 moles.