QUESTION IMAGE
Question
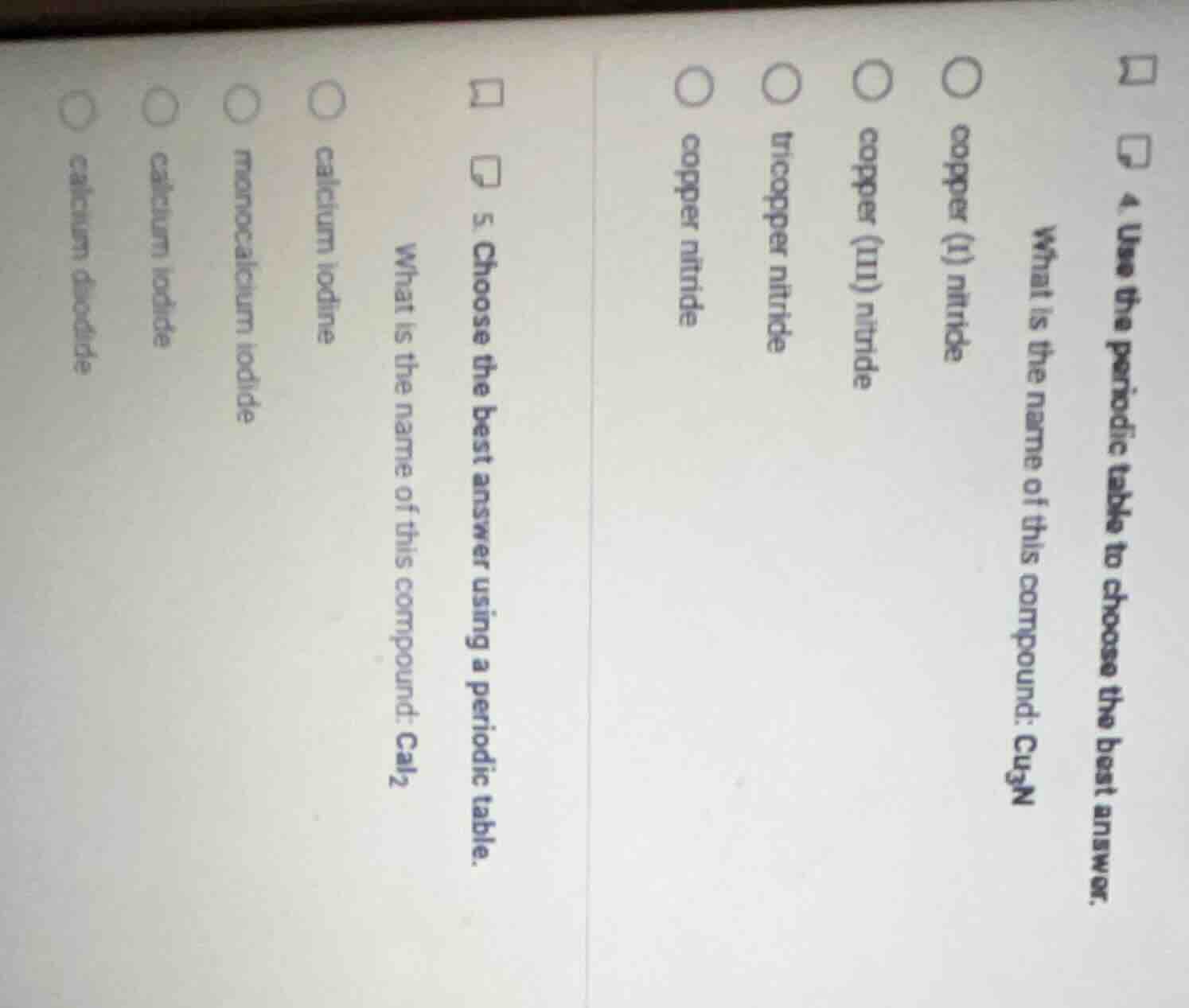
- use the periodic table to choose the best answer. what is the name of this compound: cu₃n copper (i) nitride copper (iii) nitride tricopper nitride copper nitride 5. choose the best answer using a periodic table. what is the name of this compound: cai₂ calcium iodine monocalcium iodide calcium iodide calcium diodide
Step1: Analyze $\text{Cu}_3\text{N}$ charge balance
Copper (Cu) is a transition metal; nitrogen (N) has a -3 charge. For neutral $\text{Cu}_3\text{N}$, total positive charge = +3. Each Cu has charge $\frac{+3}{3}=+1$.
Step2: Name $\text{Cu}_3\text{N}$
Transition metals need charge in parentheses. Name is copper (I) nitride.
Step3: Analyze $\text{CaI}_2$ charge balance
Calcium (Ca) is group 2, has +2 charge. Iodine (I) has -1 charge. 2 iodides balance +2 charge.
Step4: Name $\text{CaI}_2$
Group 2 metals don't need charge labels. Name is calcium iodide.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- copper (I) nitride
- calcium iodide