QUESTION IMAGE
Question
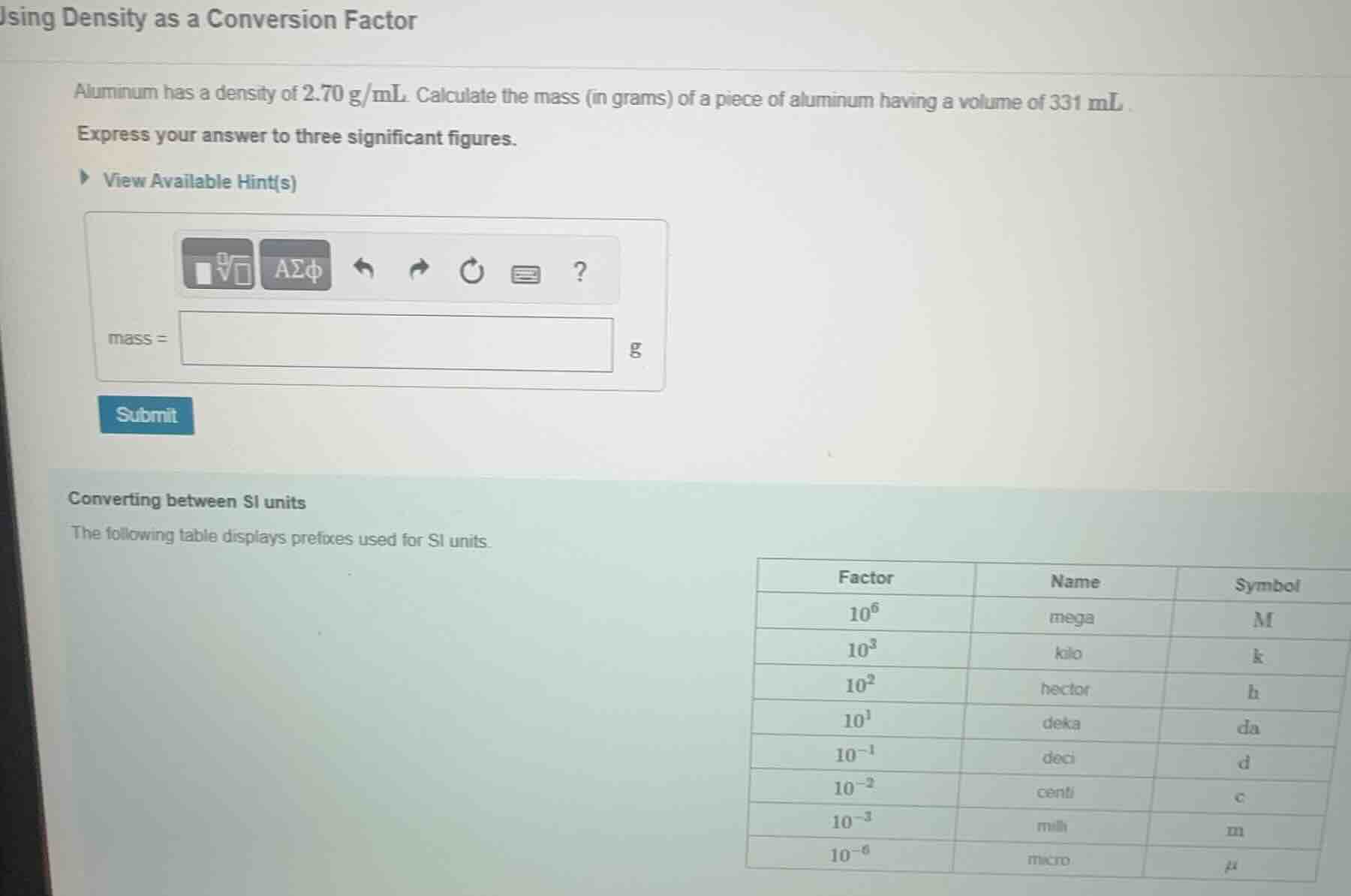
using density as a conversion factor
aluminum has a density of 2.70 g/ml. calculate the mass (in grams) of a piece of aluminum having a volume of 331 ml.
express your answer to three significant figures.
view available hint(s)
mass =
g
submit
converting between si units
the following table displays prefixes used for si units.
factor name symbol
10⁶ mega m
10³ kilo k
10² hecto h
10¹ deka da
10⁻¹ deci d
10⁻² centi c
10⁻³ milli m
10⁻⁶ micro μ
Step1: Recall density formula
Density $
ho = \frac{\text{mass }(m)}{\text{volume }(V)}$, rearrange to $m =
ho \times V$
Step2: Substitute given values
$
ho = 2.70\ \text{g/mL}$, $V = 331\ \text{mL}$
$m = 2.70\ \text{g/mL} \times 331\ \text{mL}$
Step3: Calculate and round to 3 sig figs
$m = 893.7\ \text{g} \approx 894\ \text{g}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
894