QUESTION IMAGE
Question
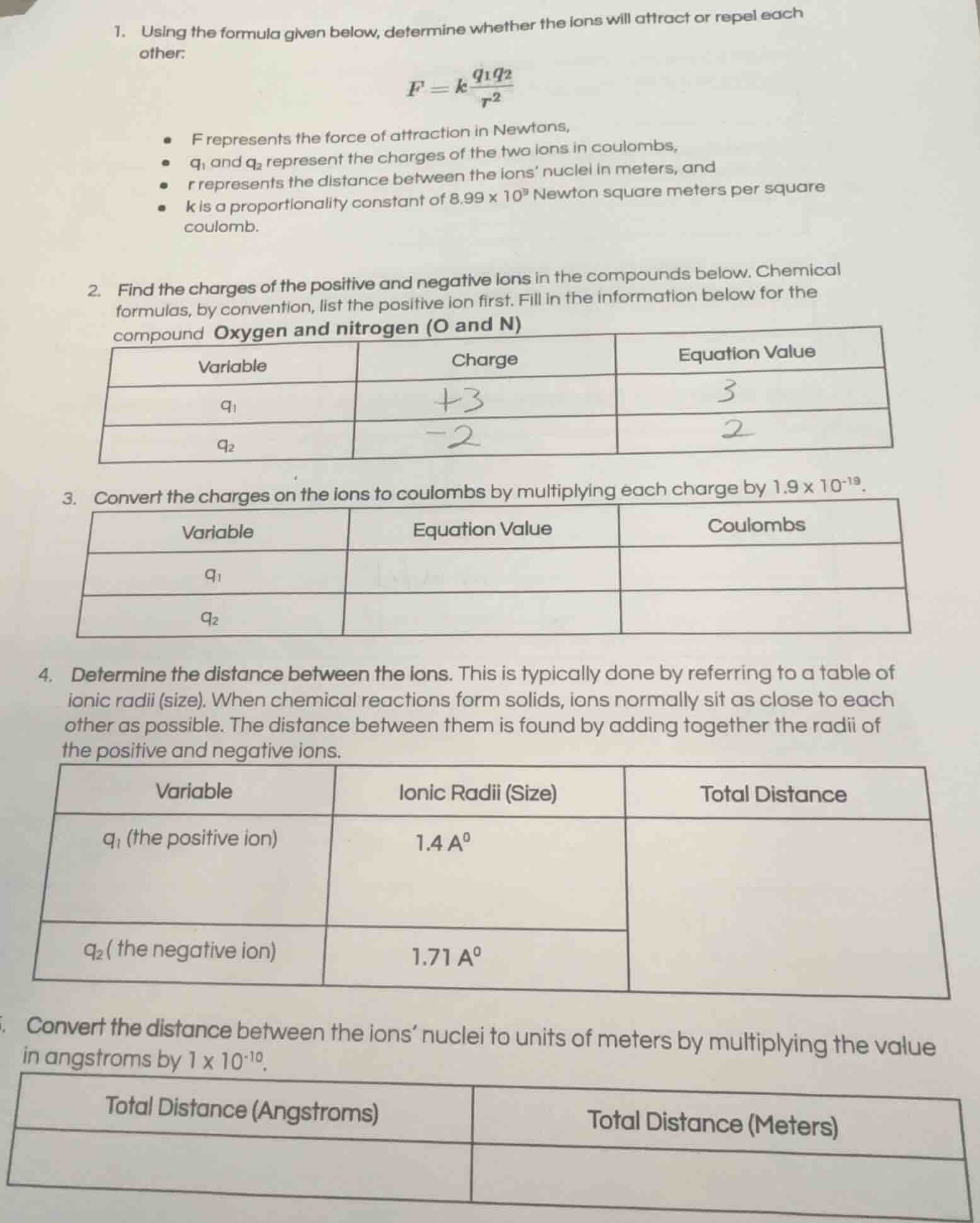
- using the formula given below, determine whether the ions will attract or repel each other.
$f = k \frac{q_1 q_2}{r^2}$
- $f$ represents the force of attraction in newtons,
- $q_1$ and $q_2$ represent the charges of the two ions in coulombs,
- $r$ represents the distance between the ions’ nuclei in meters, and
- $k$ is a proportionality constant of $8.99 \times 10^9$ newton square meters per square coulomb.
- find the charges of the positive and negative ions in the compounds below. chemical formulas, by convention, list the positive ion first. fill in the information below for the compound oxygen and nitrogen (o and n)
| variable | charge | equation value |
|---|---|---|
| $q_2$ | -2 | 2 |
- convert the charges on the ions to coulombs by multiplying each charge by $1.9 \times 10^{-19}$.
| variable | equation value | coulombs |
|---|---|---|
| $q_2$ |
- determine the distance between the ions. this is typically done by referring to a table of ionic radii (size). when chemical reactions form solids, ions normally sit as close to each other as possible. the distance between them is found by adding together the radii of the positive and negative ions.
| variable | ionic radii (size) | total distance |
|---|---|---|
| $q_2$ (the negative ion) | $1.71 \text{ a}^0$ |
- convert the distance between the ions’ nuclei to units of meters by multiplying the value in angstroms by $1 \times 10^{-10}$.
| total distance (angstroms) | total distance (meters) |
|---|
Step1: Fill ion charge values
From the given table:
$q_1 = +3$, $q_2 = -2$
Equation Value: $q_1 = 3$, $q_2 = 2$
Step2: Convert charges to coulombs
Multiply each equation value by $1.9 \times 10^{-19}$
$q_1 \text{ (Coulombs)} = 3 \times 1.9 \times 10^{-19} = 5.7 \times 10^{-19}$
$q_2 \text{ (Coulombs)} = 2 \times 1.9 \times 10^{-19} = 3.8 \times 10^{-19}$
Step3: Calculate total ionic distance
Add the two ionic radii:
$r = 1.4\ \text{Å} + 1.71\ \text{Å} = 3.11\ \text{Å}$
Step4: Convert distance to meters
Multiply angstrom value by $1 \times 10^{-10}$
$r \text{ (meters)} = 3.11 \times 1 \times 10^{-10} = 3.11 \times 10^{-10}$
Step5: Determine force sign/attraction
Substitute into Coulomb's law:
$F = k\frac{q_1 q_2}{r^2}$
Since $q_1$ is positive, $q_2$ is negative, their product is negative.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Table for Step1 (already provided, confirmed):
| Variable | Charge | Equation Value |
|---|---|---|
| $q_2$ | $-2$ | $2$ |
Table for Step2:
| Variable | Equation Value | Coulombs |
|---|---|---|
| $q_2$ | $2$ | $3.8 \times 10^{-19}$ |
Table for Step3:
| Variable | Ionic Radii (Size) | Total Distance |
|---|---|---|
| $q_2$ (negative ion) | $1.71\ \text{Å}$ | $3.11\ \text{Å}$ |
Table for Step4:
| Total Distance (Angstroms) | Total Distance (Meters) |
|---|
Final Interaction:
The ions attract each other (negative force value indicates attractive force).