QUESTION IMAGE
Question
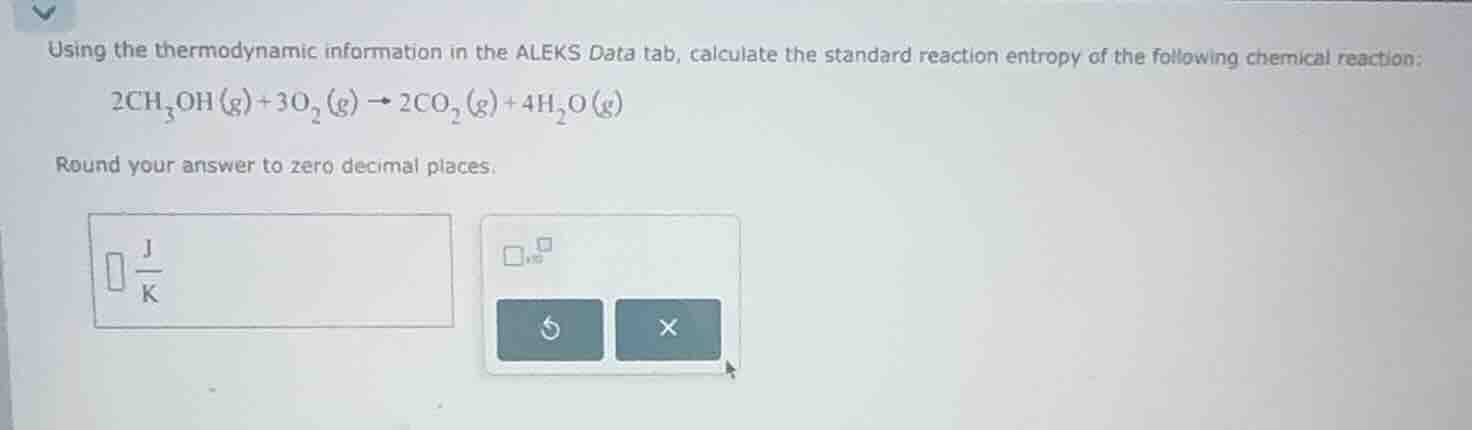
using the thermodynamic information in the aleks data tab, calculate the standard reaction entropy of the following chemical reaction: 2ch₃oh(g) + 3o₂(g) → 2co₂(g) + 4h₂o(g) round your answer to zero decimal places.
Step1: Recall the formula for standard reaction entropy
The standard reaction entropy ($\Delta S^{\circ}$) is calculated by the formula:
$\Delta S^{\circ} = \sum n \cdot S^{\circ}(\text{products}) - \sum m \cdot S^{\circ}(\text{reactants})$
where $n$ and $m$ are the stoichiometric coefficients, and $S^{\circ}$ is the standard molar entropy.
Step2: Identify standard molar entropies (from ALEKS Data or standard tables)
From thermodynamic data:
- $S^{\circ}(\text{CH}_3\text{OH}(g)) = 239.8\ \frac{\text{J}}{\text{mol·K}}$
- $S^{\circ}(\text{O}_2(g)) = 205.1\ \frac{\text{J}}{\text{mol·K}}$
- $S^{\circ}(\text{CO}_2(g)) = 213.8\ \frac{\text{J}}{\text{mol·K}}$
- $S^{\circ}(\text{H}_2\text{O}(g)) = 188.8\ \frac{\text{J}}{\text{mol·K}}$
Step3: Calculate entropy of products
For products: $2\ \text{mol}\ \text{CO}_2$ and $4\ \text{mol}\ \text{H}_2\text{O}(g)$
$\sum n \cdot S^{\circ}(\text{products}) = [2 \cdot S^{\circ}(\text{CO}_2) + 4 \cdot S^{\circ}(\text{H}_2\text{O})]$
Substitute values:
$= 2(213.8) + 4(188.8)$
$= 427.6 + 755.2$
$= 1182.8\ \frac{\text{J}}{\text{K}}$
Step4: Calculate entropy of reactants
For reactants: $2\ \text{mol}\ \text{CH}_3\text{OH}(g)$ and $3\ \text{mol}\ \text{O}_2(g)$
$\sum m \cdot S^{\circ}(\text{reactants}) = [2 \cdot S^{\circ}(\text{CH}_3\text{OH}) + 3 \cdot S^{\circ}(\text{O}_2)]$
Substitute values:
$= 2(239.8) + 3(205.1)$
$= 479.6 + 615.3$
$= 1094.9\ \frac{\text{J}}{\text{K}}$
Step5: Calculate $\Delta S^{\circ}$
$\Delta S^{\circ} = 1182.8 - 1094.9$
$\Delta S^{\circ} = 87.9\ \frac{\text{J}}{\text{K}}$ (before rounding)
Step6: Round to zero decimal places
Rounding $87.9$ to zero decimal places gives $88$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\boldsymbol{88}$