QUESTION IMAGE
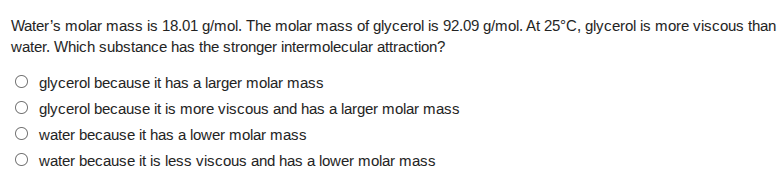
Question
waters molar mass is 18.01 g/mol. the molar mass of glycerol is 92.09 g/mol. at 25°c, glycerol is more viscous than water. which substance has the stronger intermolecular attraction?
○ glycerol because it has a larger molar mass
○ glycerol because it is more viscous and has a larger molar mass
○ water because it has a lower molar mass
○ water because it is less viscous and has a lower molar mass
Viscosity is directly related to the strength of intermolecular attractions; stronger attractions mean molecules resist flow more, increasing viscosity. A larger molar mass also correlates with stronger intermolecular forces (due to more electrons and greater London dispersion forces) in similar compounds. Since glycerol is more viscous and has a higher molar mass than water, it has stronger intermolecular attractions.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
glycerol because it is more viscous and has a larger molar mass