QUESTION IMAGE
Question
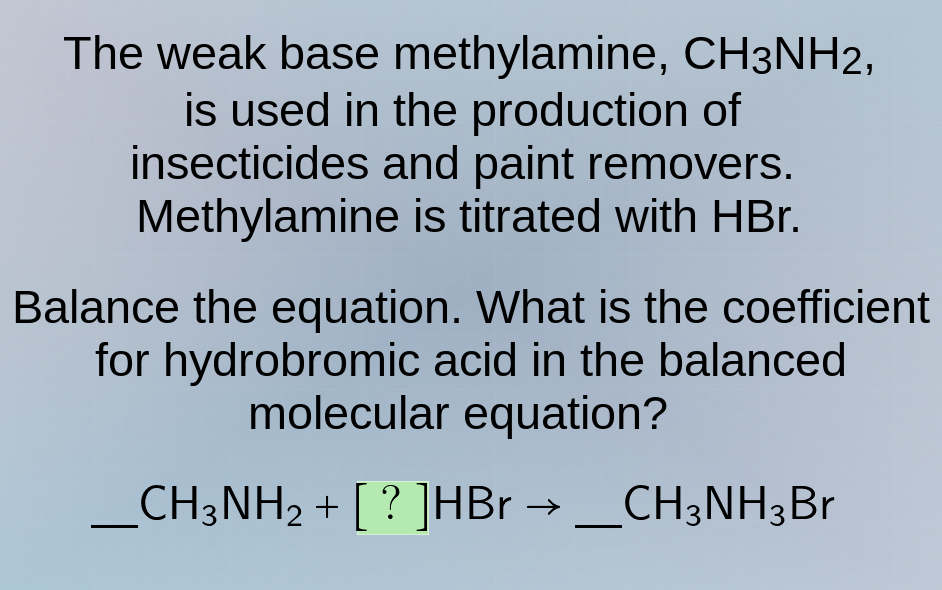
the weak base methylamine, ch₃nh₂, is used in the production of insecticides and paint removers. methylamine is titrated with hbr. balance the equation. what is the coefficient for hydrobromic acid in the balanced molecular equation? ch₃nh₂ + ?hbr → ch₃nh₃br
Step1: Analyze reactants and products
Methylamine ($\ce{CH3NH2}$) is a weak base, and hydrobromic acid ($\ce{HBr}$) is a strong acid. This is an acid - base neutralization reaction. In the product, we have $\ce{CH3NH3Br}$. Let's analyze the atoms:
- For $\ce{C}$: In $\ce{CH3NH2}$ and $\ce{CH3NH3Br}$, the number of $\ce{C}$ atoms is 1 on both sides.
- For $\ce{H}$: In $\ce{CH3NH2}$, the number of $\ce{H}$ atoms is $3 + 2=5$. In $\ce{CH3NH3Br}$, the number of $\ce{H}$ atoms is $3+3 + 1=7$ (wait, no, let's do it more carefully. $\ce{CH3NH3Br}$ has $\ce{CH3 - NH3+ - Br-}$. So $\ce{CH3}$ has 3 H, $\ce{NH3+}$ has 3 H, so total H in product is $3 + 3=6$? Wait, no, $\ce{CH3NH2}$ has formula: $\ce{C}$ (1), $\ce{H}$ (3 + 2 = 5), $\ce{N}$ (1). $\ce{HBr}$ has $\ce{H}$ (1), $\ce{Br}$ (1). $\ce{CH3NH3Br}$ has $\ce{C}$ (1), $\ce{H}$ (3+3 = 6), $\ce{N}$ (1), $\ce{Br}$ (1).
- For $\ce{N}$: In $\ce{CH3NH2}$ and $\ce{CH3NH3Br}$, the number of $\ce{N}$ atoms is 1 on both sides.
- For $\ce{Br}$: In $\ce{HBr}$ (reactant) and $\ce{CH3NH3Br}$ (product), we need to balance $\ce{Br}$. In the product, there is 1 $\ce{Br}$ atom, so in the reactant $\ce{HBr}$, we need 1 $\ce{Br}$ atom, so coefficient of $\ce{HBr}$ is related to $\ce{Br}$ balance. Also, let's consider the charge and the acid - base reaction. Methylamine ($\ce{CH3NH2}$) acts as a base, accepting a proton ($\ce{H+}$) from $\ce{HBr}$. So the reaction is: $\ce{CH3NH2 + HBr -> CH3NH3Br}$. Let's check the atoms:
- $\ce{C}$: 1 on left (in $\ce{CH3NH2}$) and 1 on right (in $\ce{CH3NH3Br}$)
- $\ce{H}$: In $\ce{CH3NH2}$, H atoms: $3 + 2=5$; in $\ce{HBr}$, H atoms: 1; total H on left: $5 + 1=6$. In $\ce{CH3NH3Br}$, H atoms: $3+3 = 6$ ( $\ce{CH3}$ has 3 H, $\ce{NH3+}$ has 3 H).
- $\ce{N}$: 1 on left and 1 on right.
- $\ce{Br}$: 1 on left (in $\ce{HBr}$) and 1 on right (in $\ce{CH3NH3Br}$).
- $\ce{C}$: 1 on left and 1 on right.
So the balanced equation is $\ce{CH3NH2 + HBr -> CH3NH3Br}$. So the coefficient for $\ce{HBr}$ is 1.
Step2: Confirm the balance
Let's write the equation with coefficients:
- Coefficient of $\ce{CH3NH2}$: 1
- Coefficient of $\ce{HBr}$: 1
- Coefficient of $\ce{CH3NH3Br}$: 1
Checking each atom:
- $\ce{C}$: 1 (left) and 1 (right)
- $\ce{H}$: $3 + 2+1=6$ (left: $\ce{CH3NH2}$ has 3 + 2 = 5 H, $\ce{HBr}$ has 1 H; total 6) and in $\ce{CH3NH3Br}$: $3+3 = 6$ H (correct)
- $\ce{N}$: 1 (left) and 1 (right)
- $\ce{Br}$: 1 (left) and 1 (right)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
1