QUESTION IMAGE
Question
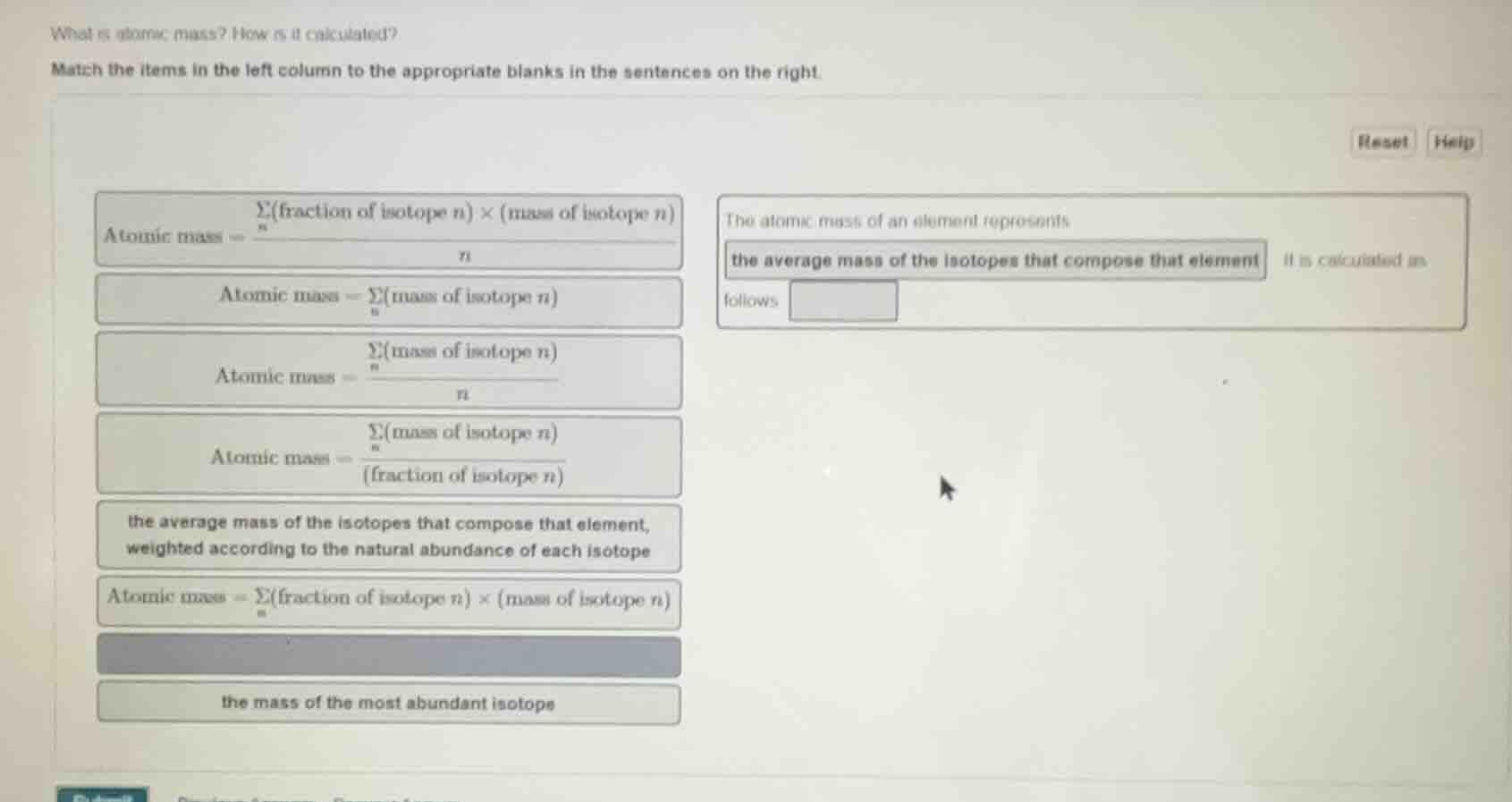
what is atomic mass? how is it calculated?
match the items in the left column to the appropriate blanks in the sentences on the right.
atomic mass = \\(\frac{\sum_{n}(\text{fraction of isotope } n) \times (\text{mass of isotope } n)}{n}\\)
atomic mass = \\(\sum_{n}(\text{mass of isotope } n)\\)
atomic mass = \\(\frac{\sum_{n}(\text{mass of isotope } n)}{n}\\)
atomic mass = \\(\frac{\sum_{n}(\text{mass of isotope } n)}{(\text{fraction of isotope } n)}\\)
the average mass of the isotopes that compose that element, weighted according to the natural abundance of each isotope
atomic mass = \\(\sum_{n}(\text{fraction of isotope } n) \times (\text{mass of isotope } n)\\)
the mass of the most abundant isotope
the atomic mass of an element represents
the average mass of the isotopes that compose that element it is calculated as follows
To determine the correct formula for calculating atomic mass, we recall that atomic mass is a weighted average of the masses of an element's isotopes, where the weighting factor is the natural abundance (fraction) of each isotope. The formula for atomic mass is the sum over each isotope of (fraction of isotope \( n \)) multiplied by (mass of isotope \( n \)). Also, the description of atomic mass should include that it's the average mass of the isotopes weighted by their natural abundances.
For the first blank (what atomic mass represents), the correct description is "the average mass of the isotopes that compose that element, weighted according to the natural abundance of each isotope" because atomic mass accounts for the relative abundance of each isotope. For the calculation formula, the correct one is \( \text{Atomic mass} = \sum_{n} (\text{fraction of isotope } n) \times (\text{mass of isotope } n) \) (or the equivalent form without the unnecessary division by \( n \) as the sum already incorporates the fractions which sum to 1 in a sense, but the key is the weighted sum).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- The atomic mass of an element represents: the average mass of the isotopes that compose that element, weighted according to the natural abundance of each isotope
- It is calculated as follows: \( \text{Atomic mass} = \sum_{n} (\text{fraction of isotope } n) \times (\text{mass of isotope } n) \) (or the boxed option with this formula or the descriptive text with the formula)
(If choosing from the left column options, the correct ones are:
- For the "represents" part: "the average mass of the isotopes that compose that element, weighted according to the natural abundance of each isotope"
- For the "calculated as" part: \( \text{Atomic mass} = \sum_{n} (\text{fraction of isotope } n) \times (\text{mass of isotope } n) \) (the option with this formula))