QUESTION IMAGE
Question
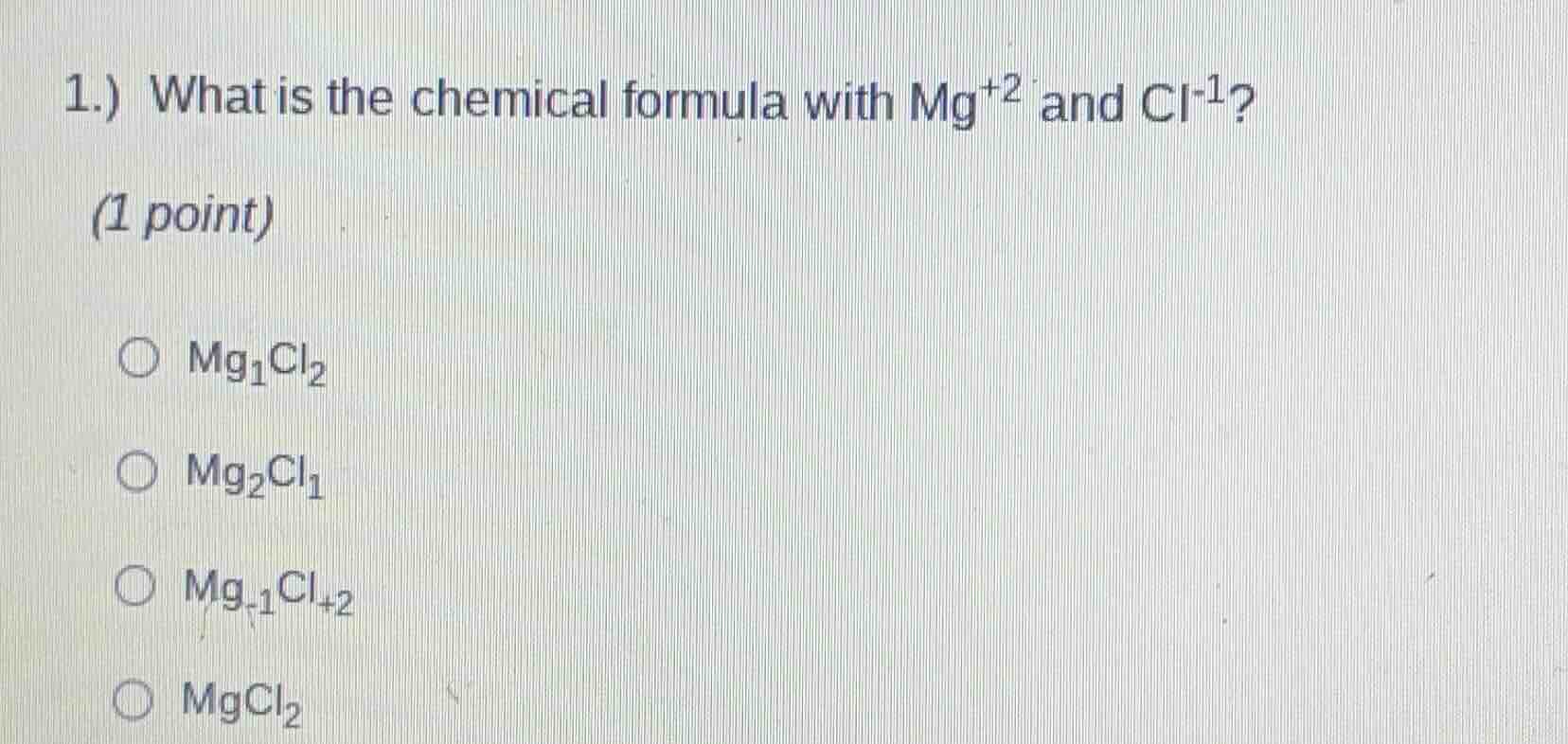
1.) what is the chemical formula with mg⁺² and cl⁻¹? (1 point) ○ mg₁cl₂ ○ mg₂cl₁ ○ mg₋₁cl₊₂ ○ mgcl₂
Step1: Recall ion combination rule
To form a neutral compound, the total positive charge equals total negative charge. \( \text{Mg}^{+2} \) has a +2 charge, \( \text{Cl}^{-1} \) has -1 charge.
Step2: Determine ion ratio
Let the number of \( \text{Mg}^{+2} \) be \( x \) and \( \text{Cl}^{-1} \) be \( y \). Then \( 2x = 1y \). The simplest ratio is \( x = 1 \), \( y = 2 \). So formula is \( \text{MgCl}_2 \) (we don't write subscript 1 for Mg).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
D. \( \text{MgCl}_2 \)