QUESTION IMAGE
Question
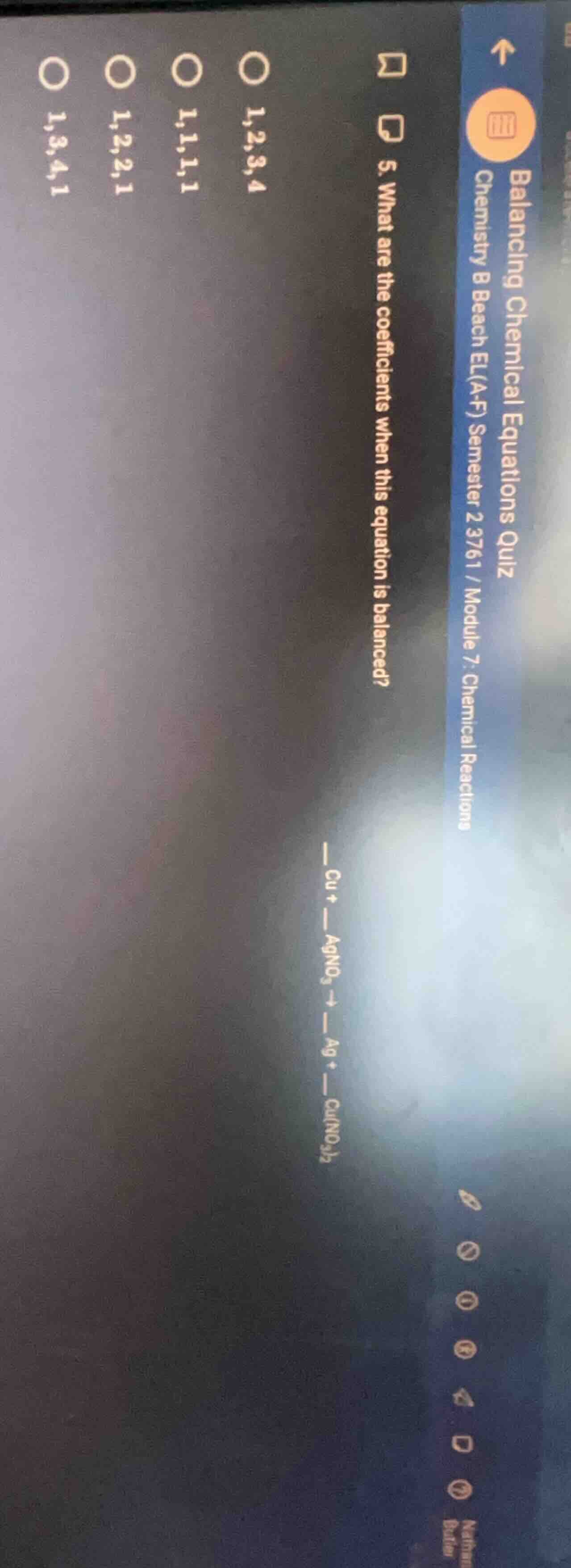
- what are the coefficients when this equation is balanced?
cu + agno₃ → ag + cu(no₃)₂
options:
1,2,3,4
1,1,1,1
1,2,2,1
1,3,4,1
Step1: Identify the unbalanced equation
The unbalanced chemical equation is: $ \text{Cu} + \text{AgNO}_3
ightarrow \text{Ag} + \text{Cu(NO}_3\text{)}_2 $
Step2: Balance the nitrate ions ($\text{NO}_3^-$)
On the right side, in $\text{Cu(NO}_3\text{)}_2$, there are 2 nitrate ions. So we need 2 moles of $\text{AgNO}_3$ on the left to balance the nitrate ions. Now the equation becomes: $ \text{Cu} + 2\text{AgNO}_3
ightarrow \text{Ag} + \text{Cu(NO}_3\text{)}_2 $
Step3: Balance the silver (Ag) atoms
Now, on the left side, we have 2 Ag atoms from $2\text{AgNO}_3$. So we need 2 moles of Ag on the right. The equation is now: $ \text{Cu} + 2\text{AgNO}_3
ightarrow 2\text{Ag} + \text{Cu(NO}_3\text{)}_2 $
Step4: Check the copper (Cu) atoms
On the left, we have 1 Cu atom, and on the right, in $\text{Cu(NO}_3\text{)}_2$, we have 1 Cu atom. So copper is already balanced.
So the coefficients are 1 (for Cu), 2 (for $\text{AgNO}_3$), 2 (for Ag), and 1 (for $\text{Cu(NO}_3\text{)}_2$), which corresponds to the option "1,2,2,1".
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
1,2,2,1 (corresponding to the option with coefficients 1,2,2,1)