QUESTION IMAGE
Question
what is the difference between a strong acid and a weak acid?
a strong acid will react faster than a weak acid in chemical reactions.
a strong acid completely dissociates in water.
a strong acid completely ionizes in water.
only a strong acid can neutralize a base.
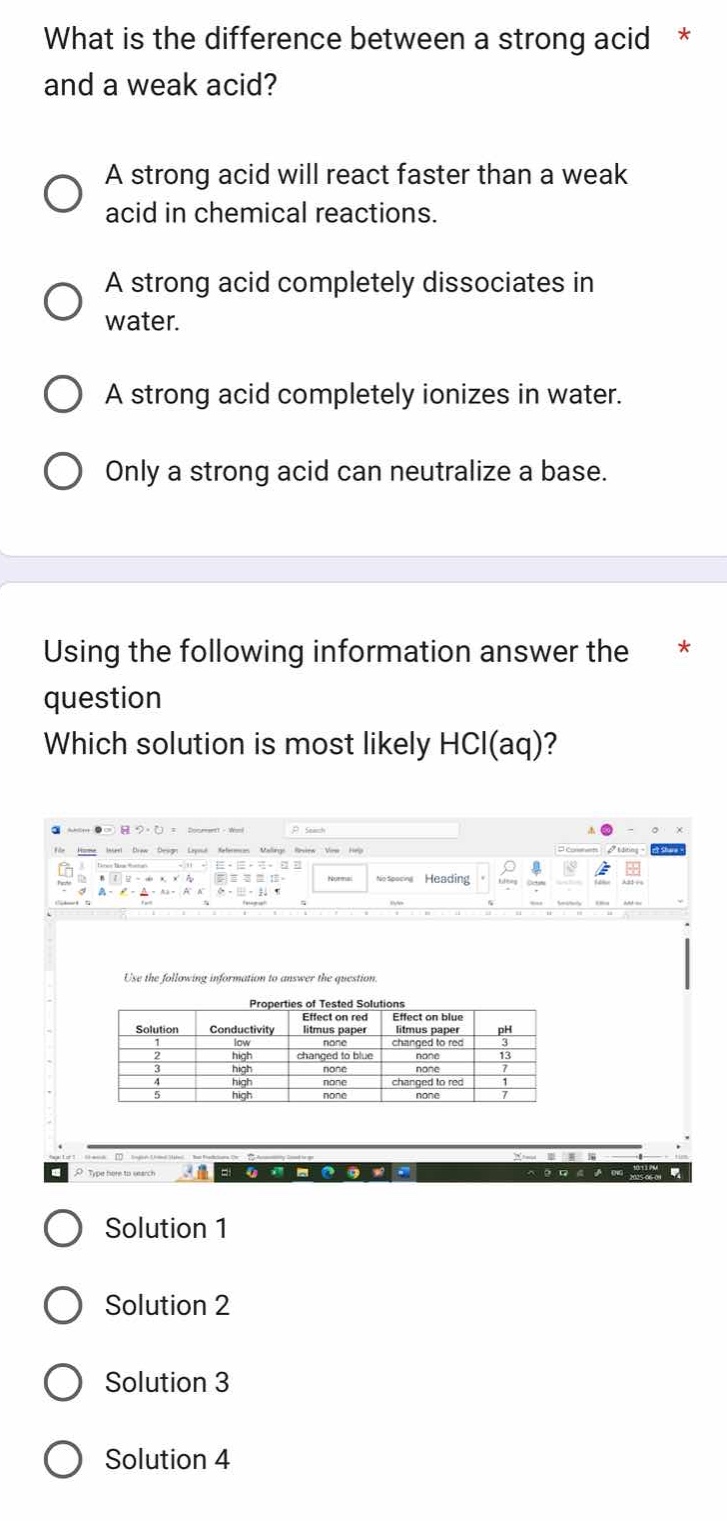
using the following information answer the question
which solution is most likely hcl(aq)?
image of a table titled properties of tested solutions with columns solution, conductivity, effect on red litmus paper, effect on blue litmus paper, ph and rows with various values
solution 1
solution 2
solution 3
solution 4
First Question:
The key distinction between strong and weak acids lies in their ionization in water. Strong acids fully break into ions, while weak acids only partially ionize. Reaction rate depends on concentration, not strength; both strong and weak acids can neutralize bases.
Step1: Recall HCl properties
HCl is a strong acid, so it has high conductivity (fully ionizes), turns blue litmus red, has low pH (<7).
Step2: Match to table data
Solution 4 has high conductivity, turns blue litmus red, pH=1 (acidic, low pH).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. A strong acid completely dissociates in water.
C. A strong acid completely ionizes in water.
---