QUESTION IMAGE
Question
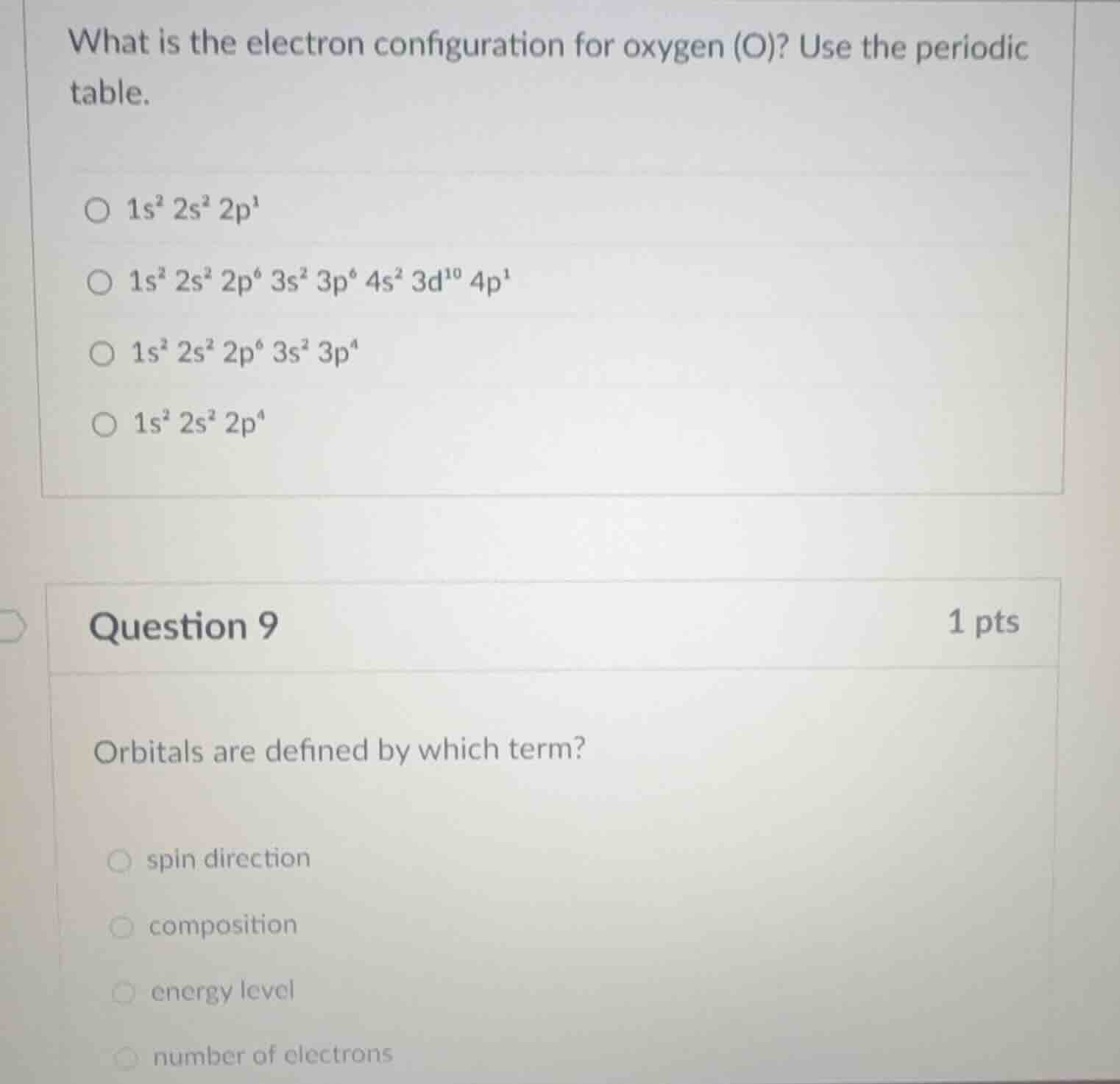
what is the electron configuration for oxygen (o)? use the periodic table. 1s² 2s² 2p¹ 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p¹ 1s² 2s² 2p⁶ 3s² 3p⁴ 1s² 2s² 2p⁴ question 9 1 pts orbitals are defined by which term? spin direction composition energy level number of electrons
First Question (Electron Configuration of Oxygen)
Oxygen has an atomic number of 8, meaning it has 8 electrons. The electron configuration follows the Aufbau principle (filling orbitals in order of increasing energy: \(1s\), \(2s\), \(2p\), etc.). The \(1s\) orbital holds 2 electrons (\(1s^2\)), \(2s\) holds 2 (\(2s^2\)), and the remaining \(8 - 2 - 2 = 4\) electrons go into the \(2p\) orbital (\(2p^4\)). So the correct configuration is \(1s^2 2s^2 2p^4\).
Orbitals are regions in an atom where electrons are likely to be found, defined by quantum numbers. The principal quantum number (\(n\)) determines the energy level (shell) of the orbital. Spin direction relates to electron spin, composition is not a term for orbital definition, and the number of electrons in an orbital is limited (e.g., \(s\) has 2, \(p\) has 6), but orbitals themselves are defined by energy level (and other quantum numbers like angular momentum, magnetic, etc.). Among the options, "energy level" is the best fit as the principal quantum number (related to energy level) is a key defining factor for an orbital's size/energy.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\(1s^2 2s^2 2p^4\) (the last option in the first question's choices)