QUESTION IMAGE
Question
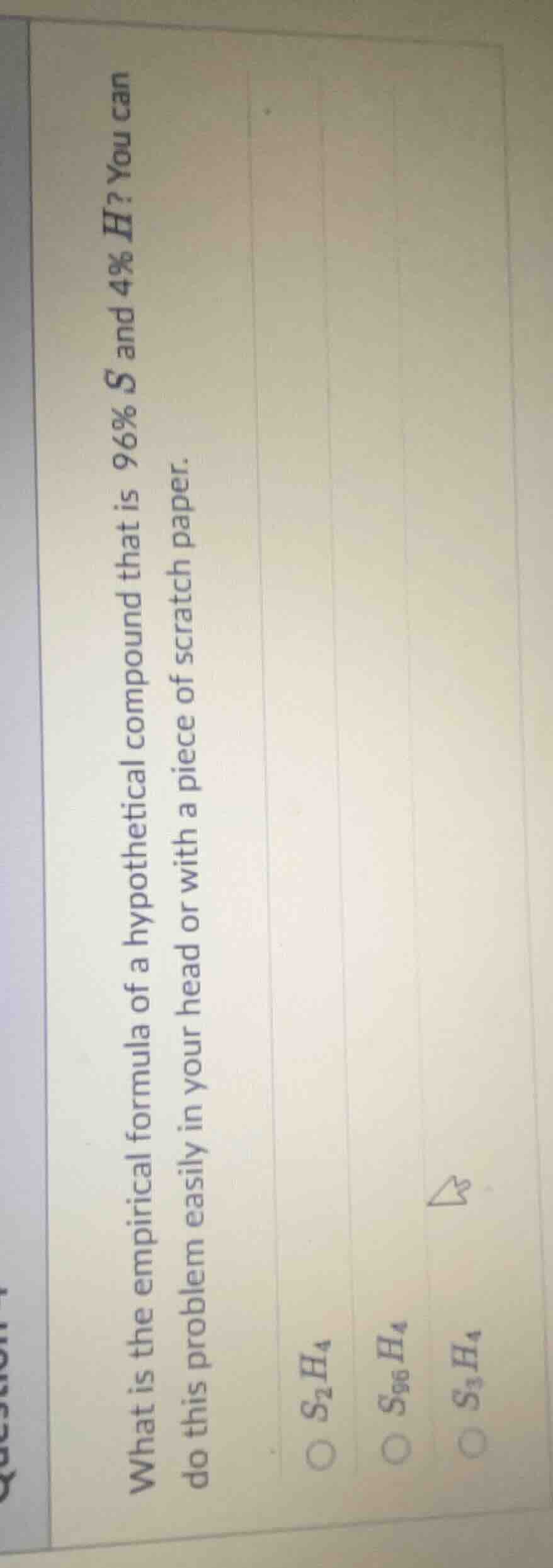
what is the empirical formula of a hypothetical compound that is 96% s and 4% h? you can do this problem easily in your head or with a piece of scratch paper. \\(\boldsymbol{s_2h_4}\\) \\(\boldsymbol{s_{96}h_4}\\) \\(\boldsymbol{s_3h_4}\\)
Step1: Assume 100g sample, get masses
Mass of S = $96\ \text{g}$, Mass of H = $4\ \text{g}$
Step2: Calculate moles of each element
Molar mass of S = $32\ \text{g/mol}$, Molar mass of H = $1\ \text{g/mol}$
Moles of S: $\frac{96\ \text{g}}{32\ \text{g/mol}} = 3\ \text{mol}$
Moles of H: $\frac{4\ \text{g}}{1\ \text{g/mol}} = 4\ \text{mol}$
Step3: Find mole ratio of S:H
Divide by smallest mole value (3 mol for S):
Ratio of S: $\frac{3}{3} = 1$
Ratio of H: $\frac{4}{3}$
Multiply both by 3 to get whole numbers: S:H = $3:4$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\boldsymbol{S_3H_4}$ (Option C)