QUESTION IMAGE
Question
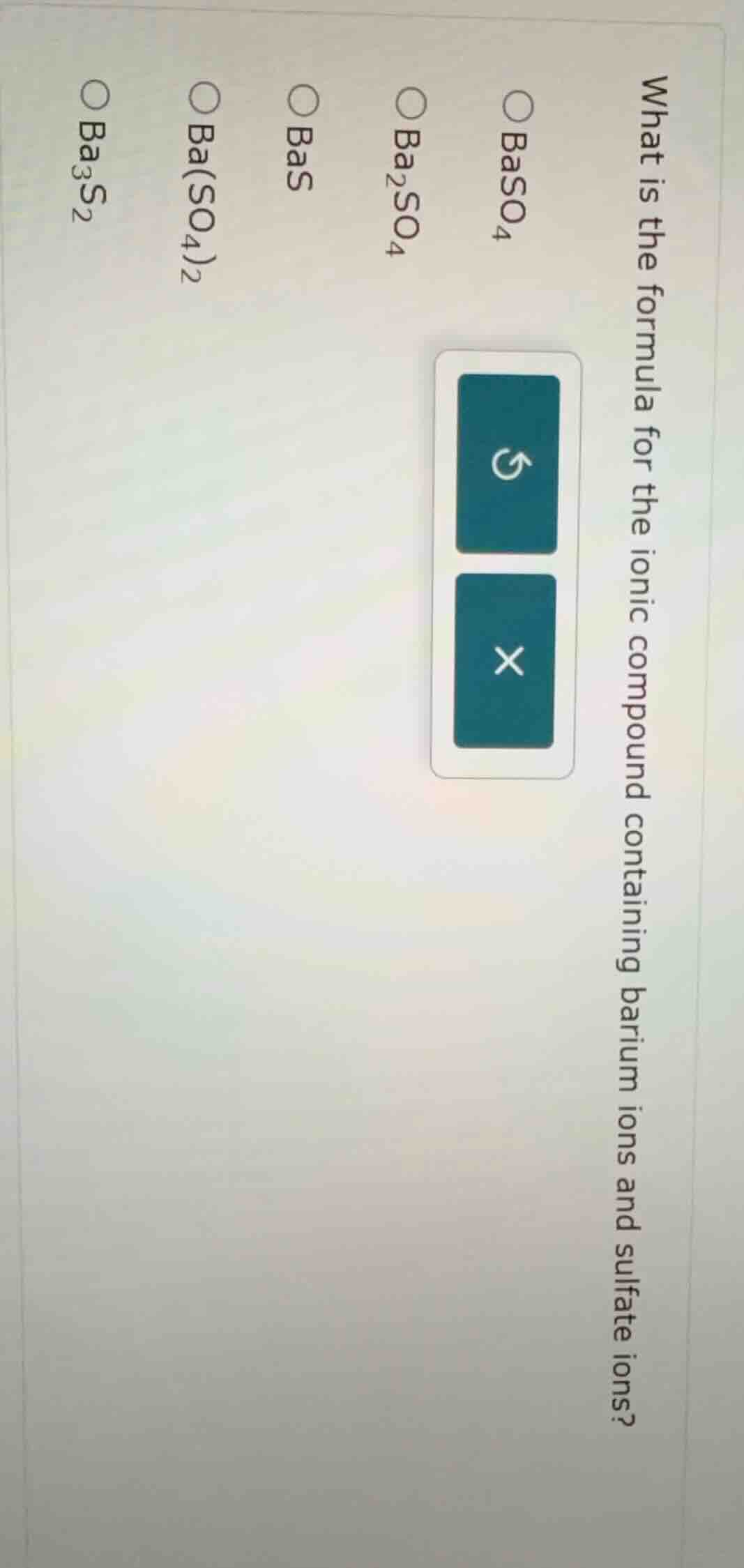
what is the formula for the ionic compound containing barium ions and sulfate ions? 〇baso₄ 〇ba₂so₄ 〇bas 〇ba(so₄)₂ 〇ba₃s₂
Step1: Determine ion charges
Barium ion is \( \text{Ba}^{2+} \), sulfate ion is \( \text{SO}_4^{2-} \).
Step2: Balance charges
To form a neutral compound, the number of positive and negative charges must be equal. Since both ions have a charge of magnitude 2, one \( \text{Ba}^{2+} \) and one \( \text{SO}_4^{2-} \) combine.
Step3: Write the formula
The formula is \( \text{BaSO}_4 \) (from the options, also check other options: \( \text{Ba}_2\text{SO}_4 \) has incorrect charge balance, \( \text{Ba(SO}_4\text{)}_2 \) would imply \( \text{Ba}^{4+} \) which is wrong, \( \text{BaS} \) and \( \text{Ba}_3\text{S}_2 \) do not involve sulfate).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. \( \text{BaSO}_4 \)