QUESTION IMAGE
Question
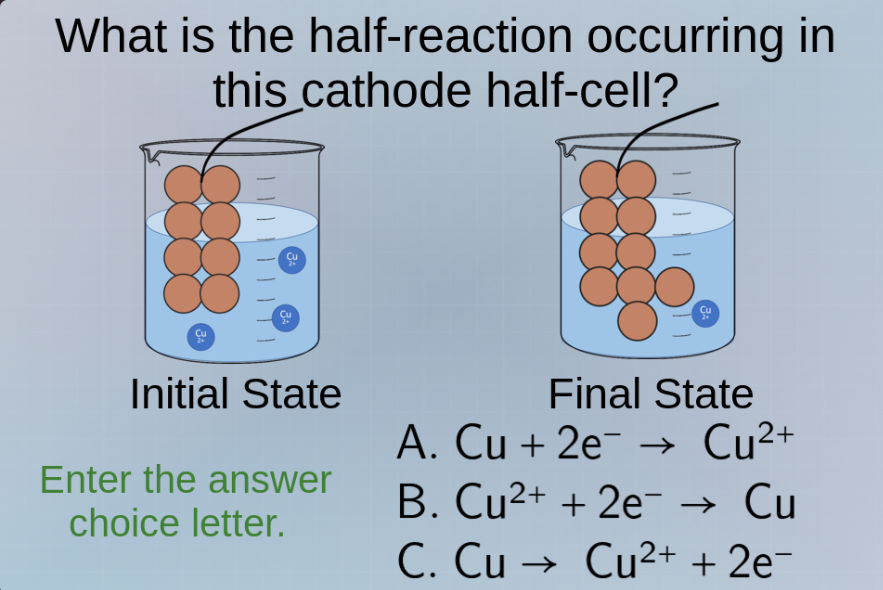
what is the half - reaction occurring in this cathode half - cell?
initial state
final state
enter the answer choice letter.
a. $\ce{cu + 2e^{-} -> cu^{2+}}$
b. $\ce{cu^{2+} + 2e^{-} -> cu}$
c. $\ce{cu -> cu^{2+} + 2e^{-}}$
In a cathode half - cell, reduction occurs (gain of electrons). Looking at the initial and final states, \(Cu^{2+}\) ions are being converted to solid Cu. The reduction half - reaction for copper (II) ions gaining electrons to form copper metal is \(Cu^{2+}+2e^-
ightarrow Cu\). Option A shows oxidation (losing electrons for Cu to form \(Cu^{2+}\)), option C is also an oxidation reaction (Cu losing electrons to form \(Cu^{2+}\)). Only option B represents a reduction reaction which occurs at the cathode.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. \( \text{Cu}^{2+} + 2\text{e}^{-}
ightarrow \text{Cu} \)