QUESTION IMAGE
Question
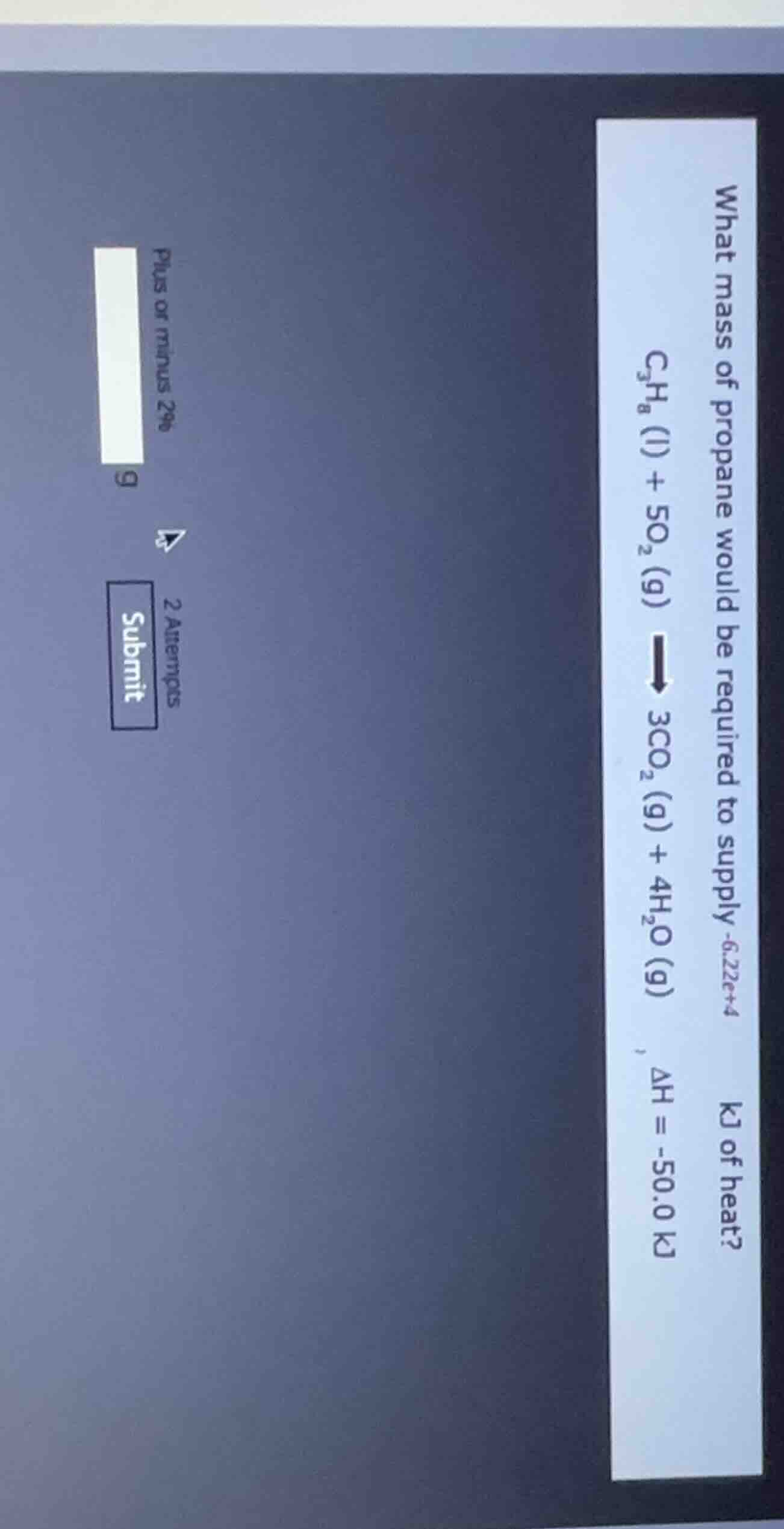
what mass of propane would be required to supply -6.22e+4 kj of heat?
c₃h₈ (l) + 5o₂ (g) → 3co₂ (g) + 4h₂o (g) δh = -50.0 kj
Step1: Find molar mass of $\text{C}_3\text{H}_8$
Molar mass = $3\times12.01 + 8\times1.008 = 44.094\ \text{g/mol}$
Step2: Calculate moles of $\text{C}_3\text{H}_8$ required
Moles = $\frac{\text{Total heat required}}{\text{Heat per mole}} = \frac{-6.22\times10^4\ \text{kJ}}{-50.0\ \text{kJ/mol}} = 1244\ \text{mol}$
Step3: Convert moles to mass
Mass = $\text{Moles} \times \text{Molar mass} = 1244\ \text{mol} \times 44.094\ \text{g/mol}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$5.48\times10^4$ g