QUESTION IMAGE
Question
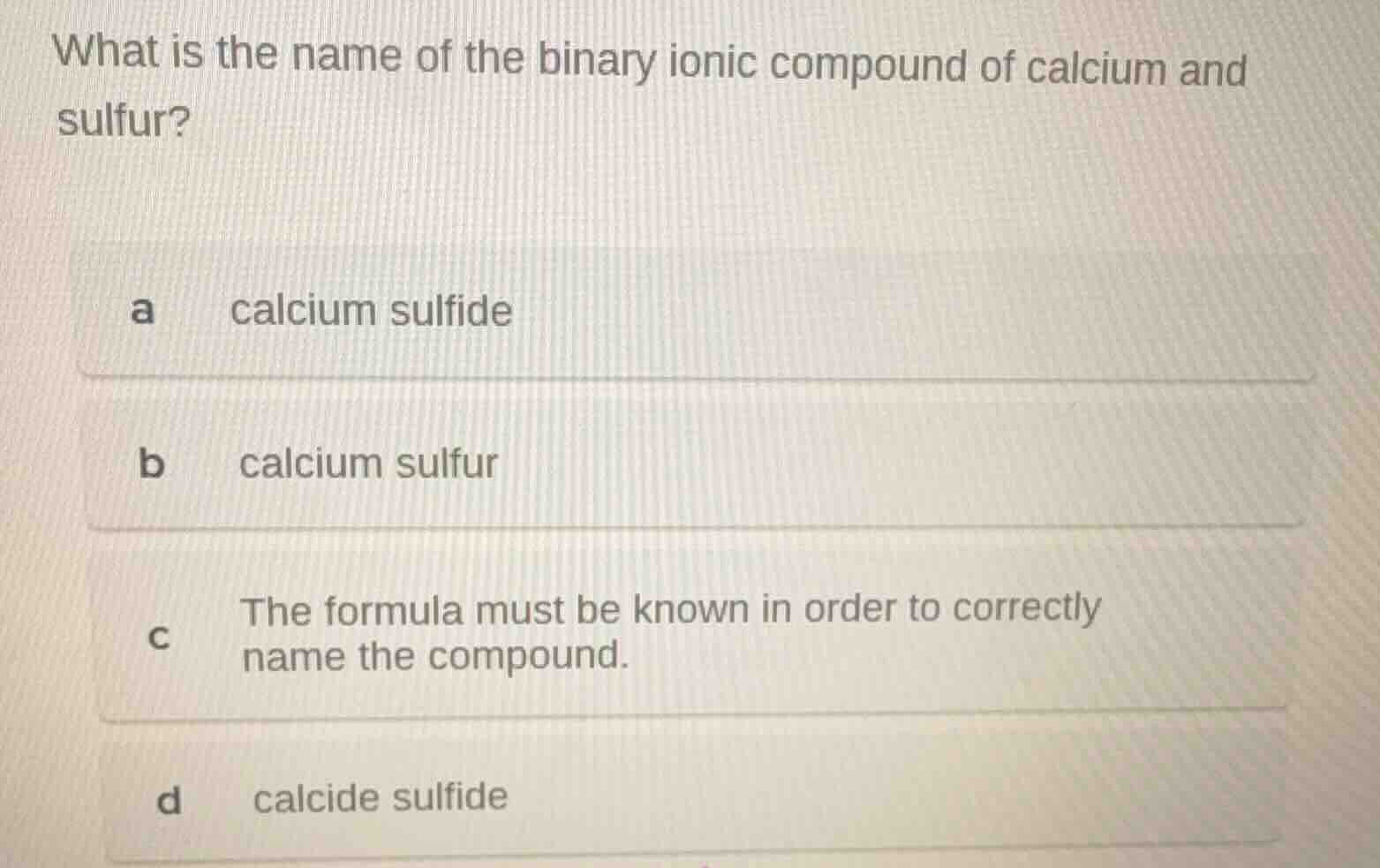
what is the name of the binary ionic compound of calcium and sulfur?
a calcium sulfide
b calcium sulfur
c the formula must be known in order to correctly name the compound.
d calcide sulfide
To name a binary ionic compound, we use the name of the cation (metal) followed by the name of the anion (non - metal) with the ending of the non - metal's name changed to - ide. Calcium (Ca) is a metal and forms the $Ca^{2 + }$ cation. Sulfur (S) is a non - metal and forms the $S^{2-}$ anion (sulfide ion). So the compound formed from calcium and sulfur is named by taking the cation name (calcium) and the anion name (sulfide). Option b is incorrect as we don't use the elemental name of the non - metal. Option c is incorrect because we can name binary ionic compounds using the cation - anion naming rules without knowing the formula (we know the charges of common ions: calcium is + 2, sulfur as an anion in binary ionic compounds is - 2, so we can determine the naming). Option d has an incorrect cation name.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
a. calcium sulfide