QUESTION IMAGE
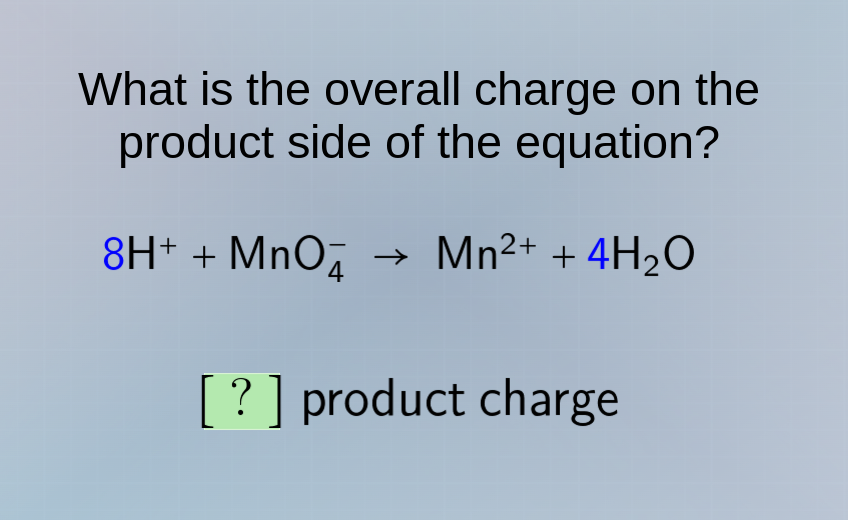
Question
what is the overall charge on the product side of the equation? 8h⁺ + mno₄⁻ → mn²⁺ + 4h₂o ? product charge
Step1: Identify charges of product species
The products are $\text{Mn}^{2+}$ and $\text{H}_2\text{O}$. $\text{H}_2\text{O}$ is neutral (charge = 0), and $\text{Mn}^{2+}$ has a charge of +2.
Step2: Sum the charges
Since there's 1 $\text{Mn}^{2+}$ and 4 $\text{H}_2\text{O}$ (which contribute 0 each), the total charge is the charge of $\text{Mn}^{2+}$: $+2 + (4\times0) = +2$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
+2