QUESTION IMAGE
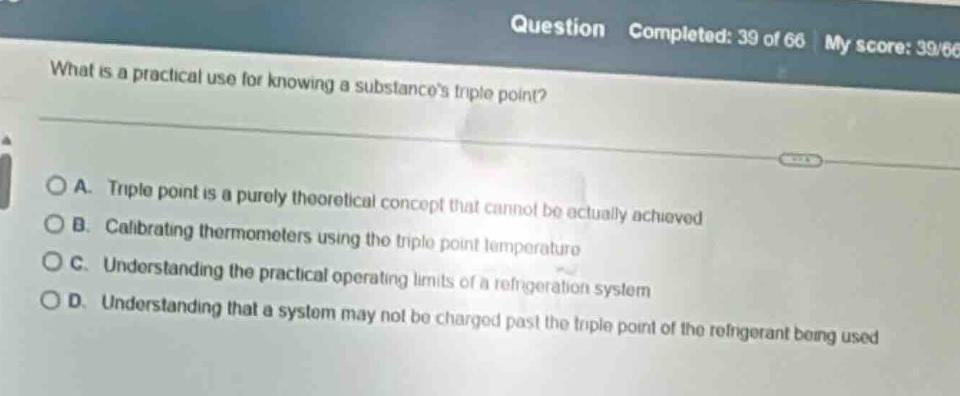
Question
what is a practical use for knowing a substances triple point?
a. triple point is a purely theoretical concept that cannot be actually achieved
b. calibrating thermometers using the triple point temperature
c. understanding the practical operating limits of a refrigeration system
d. understanding that a system may not be charged past the triple point of the refrigerant being used
The triple point of a substance (like water, with a defined triple point of 273.16 K and 611.657 Pa) is a precise, reproducible physical condition. This makes it an international standard for calibrating thermometers to ensure accurate temperature measurements. Option A is incorrect because triple points can be achieved in laboratory settings. Options C and D are not primary practical uses of knowing a substance's triple point.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. Calibrating thermometers using the triple point temperature