QUESTION IMAGE
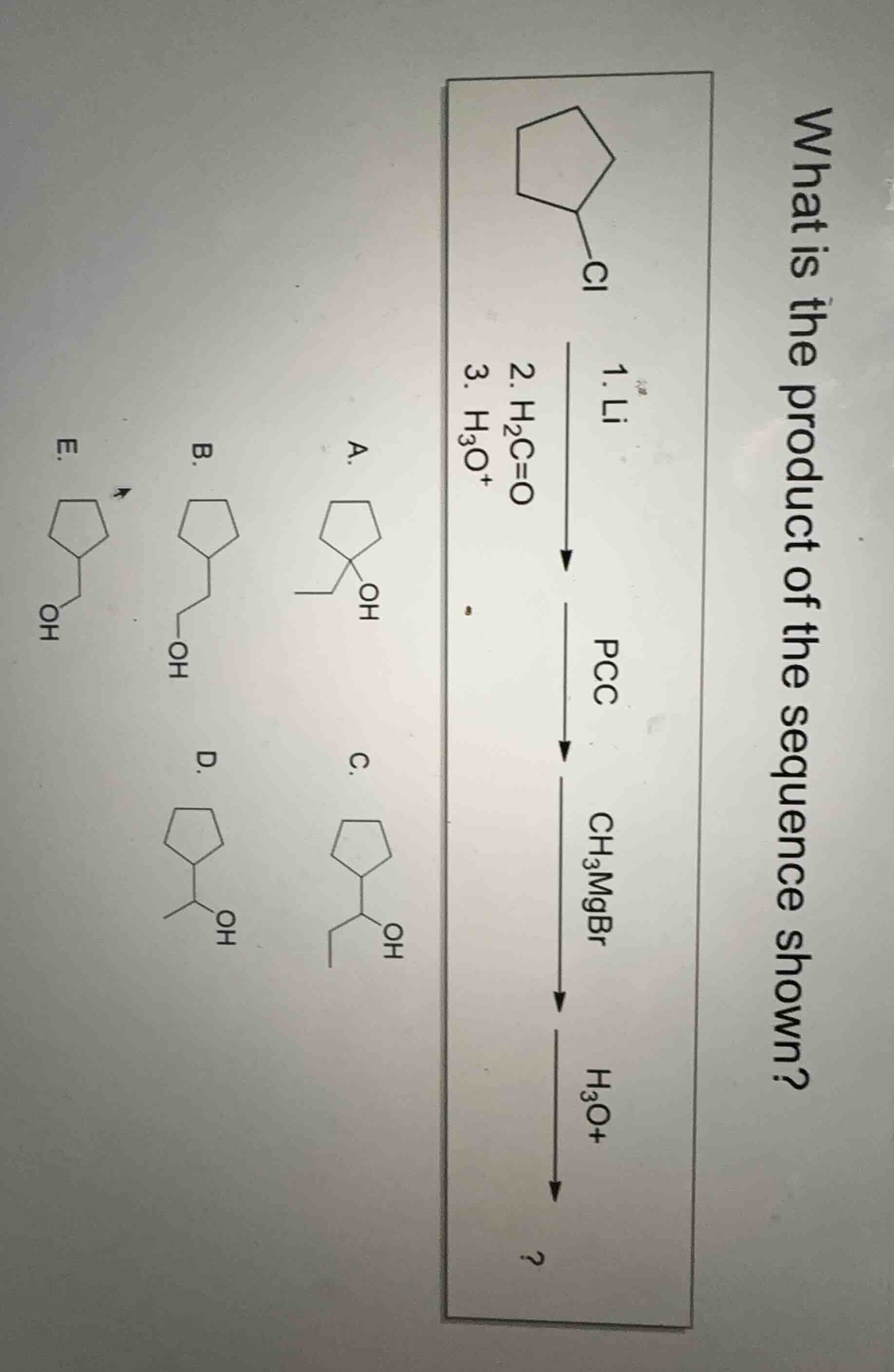
Question
what is the product of the sequence shown?
(chemical reaction sequence and options a - e with chemical structures)
Step1: Analyze the first reaction (Li, then H₂C=O, then H₃O⁺)
The starting material is chlorocyclopentane. First, with Li, it forms a cyclopentyl lithium (organolithium reagent). Then, reacting with formaldehyde (H₂C=O, an aldehyde) followed by protonation (H₃O⁺) will add a -CH₂OH group to the cyclopentyl ring. So the product after this step is cyclopentylmethanol (structure E? Wait, no, let's recheck. Wait, Li reacts with R - Cl to form R - Li. Then R - Li reacts with H₂C=O (formaldehyde) to form R - CH₂ - O⁻Li⁺, then protonation gives R - CH₂OH. So that's a primary alcohol: cyclopentylmethanol (structure E? Wait, structure E is cyclopentyl - CH₂OH? Wait, the options: E has - CH₂OH, B has - CH₂CH₂OH, A has a tertiary alcohol, C has a secondary alcohol with an ethyl group? Wait, maybe I messed up. Wait, next step is PCC (Pyridinium chlorochromate), which oxidizes primary alcohols to aldehydes, secondary to ketones. So cyclopentylmethanol (primary alcohol) with PCC will be oxidized to cyclopentanecarbaldehyde (cyclopentyl - CHO). Then, reacting with CH₃MgBr (Grignard reagent) and then H₃O⁺: Grignard adds a CH₃ group to the carbonyl carbon. So cyclopentanecarbaldehyde (CHO group) + CH₃MgBr → after protonation, it's cyclopentyl - CH(OH) - CH₃? Wait, no: aldehyde (R - CHO) + RMgX → R - CH(OH) - R'. So here, R is cyclopentyl, R' is CH₃. So the product should be cyclopentyl - CH(OH) - CH₃? Wait, no, wait: cyclopentanecarbaldehyde is C₅H₉ - CHO. Adding CH₃MgBr: the Grignard attacks the carbonyl, forming a secondary alcohol: C₅H₉ - CH(OH) - CH₃? Wait, but let's check the options. Option D: cyclopentyl - CH(OH) - CH₃? Wait, option C: cyclopentyl - CH(OH) - CH₂CH₃? No, option C has an ethyl group? Wait, maybe I made a mistake in the first step. Wait, first reaction: Li with chlorocyclopentane: C₅H₉ - Cl + Li → C₅H₉ - Li (organolithium). Then H₂C=O (formaldehyde) → C₅H₉ - CH₂ - O⁻Li⁺, then H₃O⁺ → C₅H₉ - CH₂OH (primary alcohol, cyclopentylmethanol). Then PCC oxidizes primary alcohol to aldehyde: C₅H₉ - CHO (cyclopentanecarbaldehyde). Then CH₃MgBr (Grignard) adds CH₃ to the carbonyl: C₅H₉ - CH(OH) - CH₃ (secondary alcohol, with a methyl group). Wait, option D is cyclopentyl - CH(OH) - CH₃? Wait, option D's structure: cyclopentyl attached to a carbon with OH and CH₃? Yes. Wait, but let's re - evaluate the steps:
- Step 1: Formation of organolithium and reaction with formaldehyde
- \( \text{Cyclopentyl - Cl} + \text{Li}
ightarrow \text{Cyclopentyl - Li} \) (organolithium reagent).
- \( \text{Cyclopentyl - Li} + \text{H}_2\text{C}=\text{O}
ightarrow \text{Cyclopentyl - CH}_2\text{ - O}^-\text{Li}^+ \) (after nucleophilic attack on formaldehyde).
- \( \text{Cyclopentyl - CH}_2\text{ - O}^-\text{Li}^+ + \text{H}_3\text{O}^+
ightarrow \text{Cyclopentyl - CH}_2\text{OH} \) (primary alcohol, cyclopentylmethanol).
- Step 2: Oxidation with PCC
- Primary alcohol (\( \text{Cyclopentyl - CH}_2\text{OH} \)) is oxidized by PCC to aldehyde: \( \text{Cyclopentyl - CHO} \) (cyclopentanecarbaldehyde).
- Step 3: Reaction with Grignard reagent (CH₃MgBr) and protonation
- Aldehyde (\( \text{Cyclopentyl - CHO} \)) reacts with \( \text{CH}_3\text{MgBr} \): the Grignard reagent (\( \text{CH}_3\text{MgBr} \)) is a nucleophile that attacks the carbonyl carbon of the aldehyde.
- The intermediate after attack is \( \text{Cyclopentyl - CH(OMgBr) - CH}_3 \), and after protonation (\( \text{H}_3\text{O}^+ \)) it becomes \( \text{Cyclopentyl - CH(OH) - CH}_3 \), which is a secondary alcohol with a methyl group attached to the carbon bearing…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
D. \(\boldsymbol{\text{Cyclopentyl - CH(OH) - CH}_3}\) (the structure corresponding to option D)