QUESTION IMAGE
Question
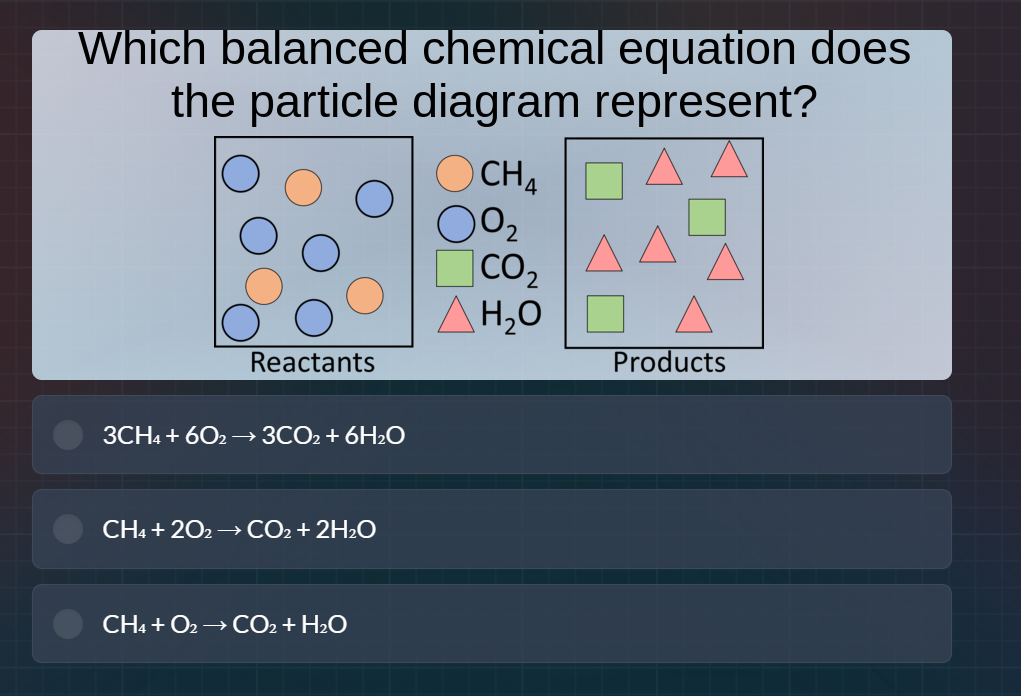
which balanced chemical equation does the particle diagram represent? reactants: 3 ch₄ (orange), 6 o₂ (blue); products: 3 co₂ (green), 6 h₂o (pink). options: 3ch₄ + 6o₂ → 3co₂ + 6h₂o; ch₄ + 2o₂ → co₂ + 2h₂o; ch₄ + o₂ → co₂ + h₂o
Step1: Count reactant particles
- \( \text{CH}_4 \) (orange): 3 particles.
- \( \text{O}_2 \) (blue): 6 particles.
Step2: Count product particles
- \( \text{CO}_2 \) (green): 3 particles.
- \( \text{H}_2\text{O} \) (pink): 6 particles.
Step3: Match to balanced equation
The counts match the equation \( 3\text{CH}_4 + 6\text{O}_2
ightarrow 3\text{CO}_2 + 6\text{H}_2\text{O} \), as 3 \( \text{CH}_4 \), 6 \( \text{O}_2 \) produce 3 \( \text{CO}_2 \) and 6 \( \text{H}_2\text{O} \). The second option has 1 \( \text{CH}_4 \), 2 \( \text{O}_2 \), 1 \( \text{CO}_2 \), 2 \( \text{H}_2\text{O} \) (doesn't match counts). The third is unbalanced.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. \( 3\text{CH}_4 + 6\text{O}_2
ightarrow 3\text{CO}_2 + 6\text{H}_2\text{O} \)