QUESTION IMAGE
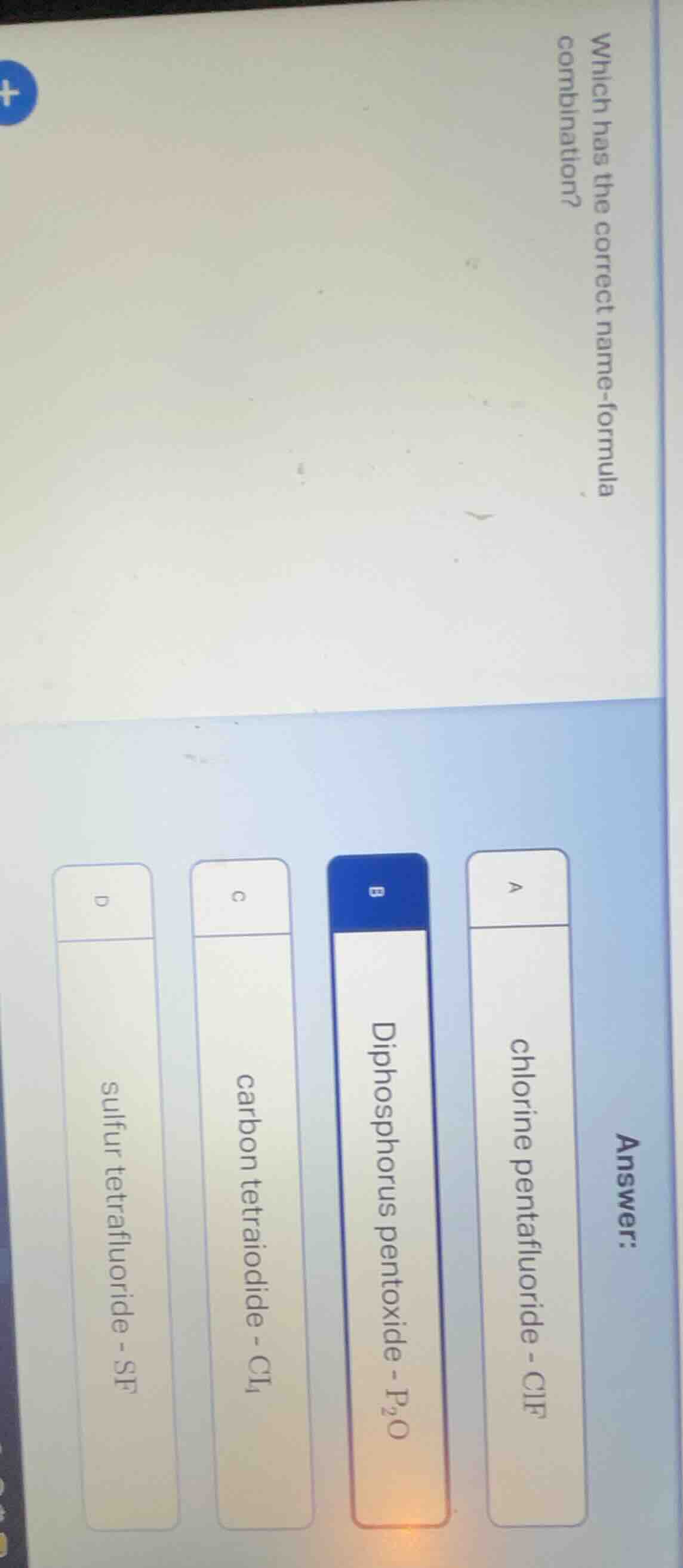
Question
which has the correct name-formula combination?
answer:
a chlorine pentafluoride - clf
b diphosphorus pentoxide - $p_2o$
c carbon tetraiodide - $ci_4$
d sulfur tetrafluoride - sf
Brief Explanations
- Option A: "Chlorine pentafluoride" requires 5 fluorine atoms, so the formula should be $\text{ClF}_5$, not $\text{ClF}$.
- Option B: "Diphosphorus pentoxide" means 2 phosphorus and 5 oxygen atoms, so the formula should be $\text{P}_2\text{O}_5$, not $\text{P}_2\text{O}$.
- Option C: "Carbon tetraiodide" means 1 carbon and 4 iodine atoms, so the formula should be $\text{CI}_4$, which matches.
- Option D: "Sulfur tetrafluoride" requires 4 fluorine atoms, so the formula should be $\text{SF}_4$, not $\text{SF}$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. carbon tetraiodide - $\text{CI}_4$