QUESTION IMAGE
Question
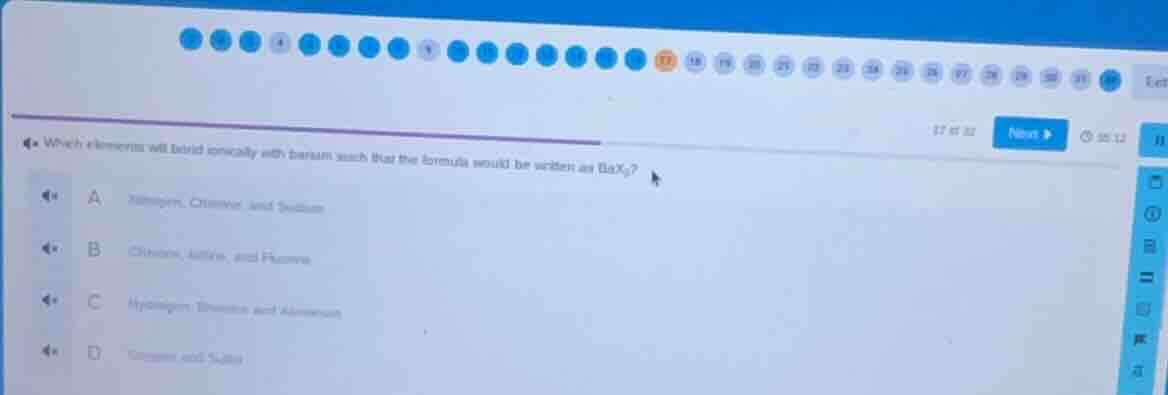
which elements will bond ionically with barium such that the formula would be written as bax₂?
a nitrogen, chlorine, and sodium
b chlorine, iodine, and fluorine
c hydrogen, bromine, and aluminum
d oxygen and sulfur
Barium (Ba) is a group 2 element, so it forms a +2 cation ($\text{Ba}^{2+}$). For the formula $\text{BaX}_2$, each X ion must have a -1 charge to balance the +2 charge of Ba. Group 17 (halogen) elements form -1 anions. Chlorine, iodine, and fluorine are all halogens that form -1 ions, so they will bond with barium to make $\text{BaX}_2$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. Chlorine, Iodine, and Fluorine