QUESTION IMAGE
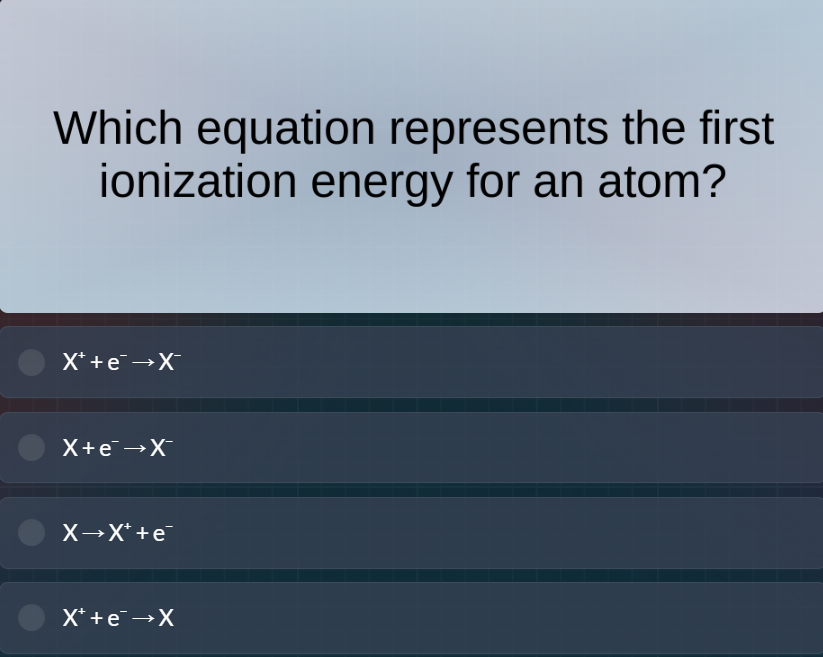
Question
which equation represents the first ionization energy for an atom?
$x^++e^- \
ightarrow x^-$
$x+e^- \
ightarrow x^-$
$x \
ightarrow x^++e^-$
$x^++e^- \
ightarrow x$
Brief Explanations
First ionization energy is defined as the energy required to remove one electron from a neutral gaseous atom, producing a positively charged ion and a free electron. We match this definition to the correct reaction.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
X→X⁺+e⁻