QUESTION IMAGE
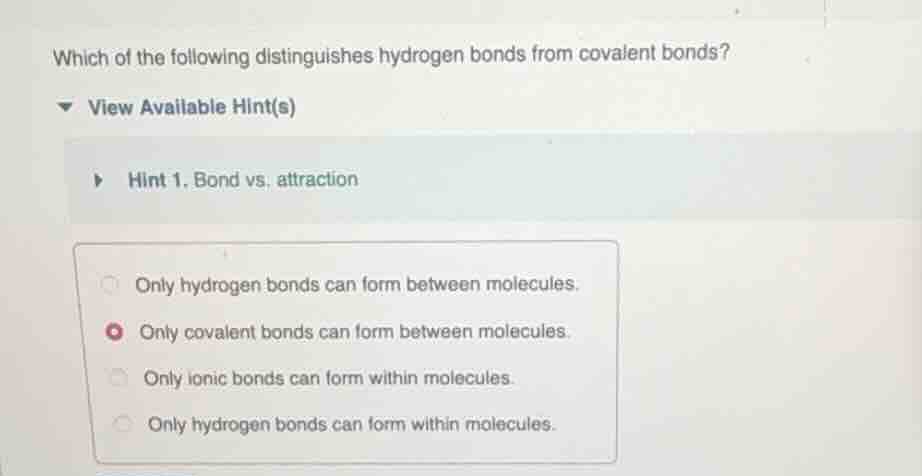
Question
which of the following distinguishes hydrogen bonds from covalent bonds? view available hint(s) hint 1. bond vs. attraction only hydrogen bonds can form between molecules. only covalent bonds can form between molecules. only ionic bonds can form within molecules. only hydrogen bonds can form within molecules.
Covalent bonds are intramolecular forces that form within molecules by sharing electrons between atoms. Hydrogen bonds are intermolecular forces that form between molecules (between a hydrogen atom bonded to O/N/F in one molecule and an O/N/F atom in another). The incorrect options are invalid: covalent bonds do not form between molecules, ionic bonds are intramolecular, and hydrogen bonds do not form within molecules.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Only hydrogen bonds can form between molecules.