QUESTION IMAGE
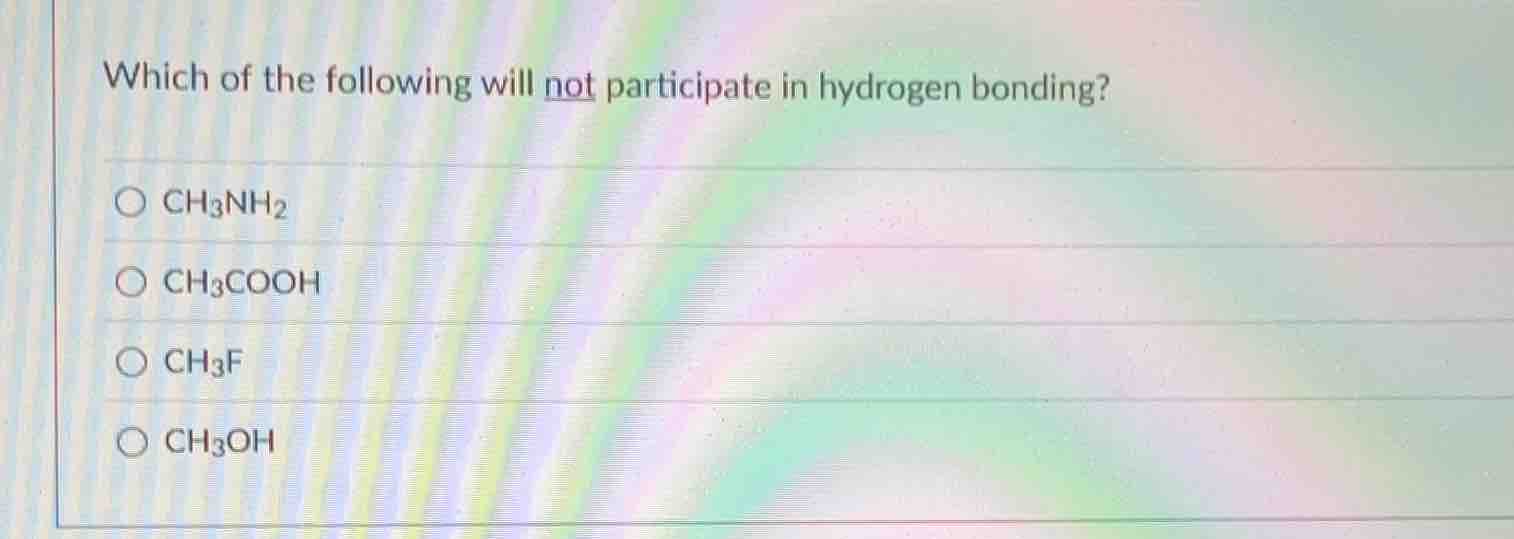
Question
which of the following will not participate in hydrogen bonding?\
\\(\bigcirc\\) \\(\ce{ch3nh2}\\)\
\\(\bigcirc\\) \\(\ce{ch3cooh}\\)\
\\(\bigcirc\\) \\(\ce{ch3f}\\)\
\\(\bigcirc\\) \\(\ce{ch3oh}\\)
To determine which compound does not participate in hydrogen bonding, we recall the conditions for hydrogen bonding: a hydrogen atom must be bonded to a highly electronegative atom (N, O, or F), and there must be a lone pair on N, O, or F in another molecule (or the same molecule for intramolecular H - bonding).
- For \( \text{CH}_3\text{NH}_2 \): The \( \text{N} \) has lone pairs, and \( \text{H} \) is bonded to \( \text{N} \), so it can form H - bonds.
- For \( \text{CH}_3\text{COOH} \): The \( \text{O} \) in \( -\text{COOH} \) has lone pairs, and \( \text{H} \) is bonded to \( \text{O} \), so it can form H - bonds (both intramolecular and intermolecular).
- For \( \text{CH}_3\text{F} \): The \( \text{H} \) in \( \text{CH}_3\text{F} \) is bonded to \( \text{C} \), not to \( \text{F} \) (since \( \text{C} - \text{H} \) bonds are not polar enough for H - bonding with \( \text{F} \) in terms of the H - bonding requirement that H is bonded to N, O, or F). The \( \text{F} \) has lone pairs, but since the H is not bonded to F (or N, O), it cannot form H - bonds.
- For \( \text{CH}_3\text{OH} \): The \( \text{O} \) has lone pairs, and \( \text{H} \) is bonded to \( \text{O} \), so it can form H - bonds.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\( \boldsymbol{\text{CH}_3\text{F}} \) (the option with \( \text{CH}_3\text{F} \))